The molecule that has a linear structure is:
(A). $C{O_2}$
(B). $N{O_2}$
(C). $S{O_2}$
(D). $Si{O_2}$
Answer
597k+ views
Hint: The three dimensional shapes of the many small molecules are often predicted by applying the valence shell electron pair repulsion theory (VSEPR) when atoms combine to make molecules, pair of valence electrons arrange themselves as far away from one another as possible. Differently to characterize molecular shape is in terms of hybrid orbitals.
Complete answer:
Linear molecules may be molecules during which atoms are deployed in a straight line (under $180^\circ $ ange.) have $sp$ hybridization at the central atom .
For example $ \Rightarrow C{O_2}$
$O = C = O$
Linear structure
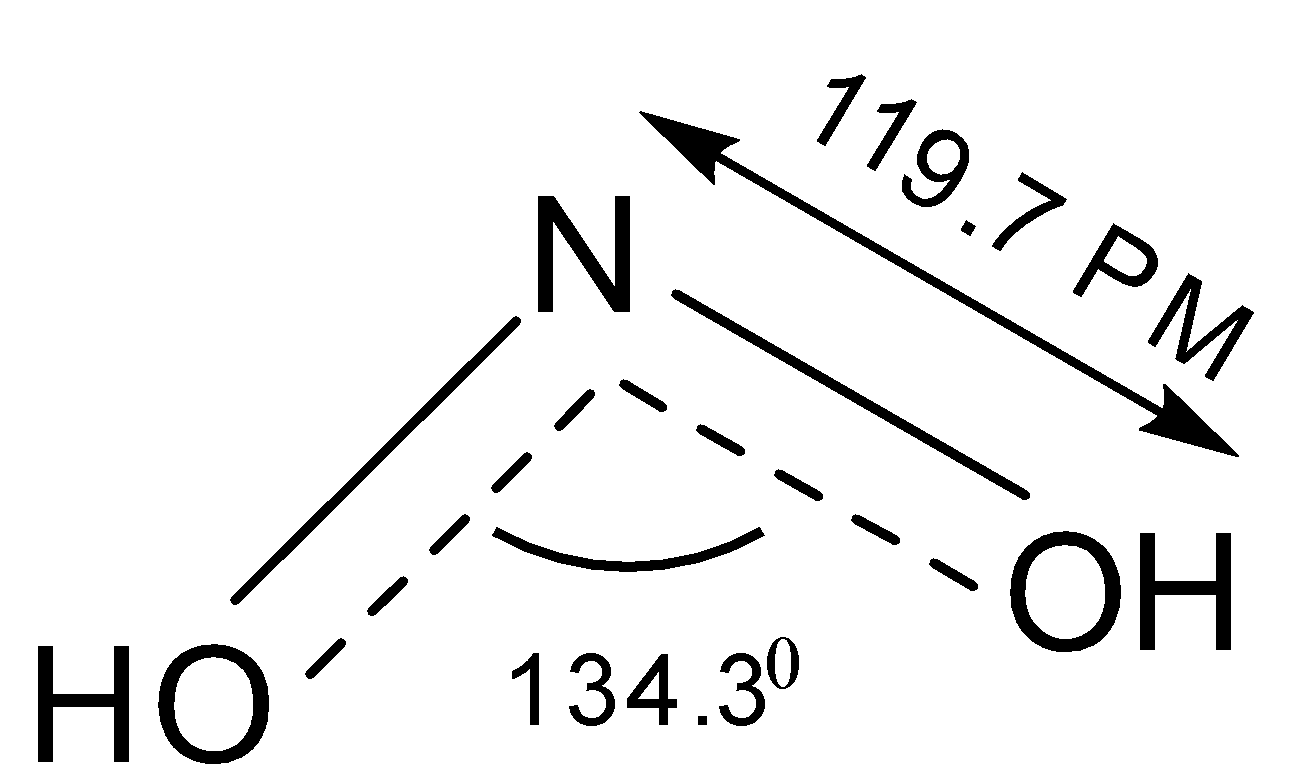
-$N{O_2} \Rightarrow $ It has bent shape
-The molecular shape of $S{O_2}$ is V-shaped or bent shape.

-$Si{O_2}$ , silicon atom shows tetrahedral coordination, with four oxygen atoms surrounding a central silicon atom
Hence option A is correct .
Additional information :
Though VSEPR theory is able to explain the shapes of simple molecules but it is unable to predict the shape in a number of cases . Thus , it had a limited application only . Moreover , talking of the direction of electron pairs does not seem to be very rational . Thus , Lewis' approach as well as VSEPR theory had a number of limitations .
To explain the above limitations , two important theories called modern theories of covalent bond formation have been put forward , that is the valence bond theory and the molecular orbital theory .
Note:
While predicting the geometry of molecules containing double bonds , it should be kept in mind that the double bond is considered one electron pair . For example , in case of ozone the central oxygen atom is considered to have two bond pairs and one lone pair and has a bent shape structure .
Complete answer:
Linear molecules may be molecules during which atoms are deployed in a straight line (under $180^\circ $ ange.) have $sp$ hybridization at the central atom .
For example $ \Rightarrow C{O_2}$
$O = C = O$
Linear structure
-$N{O_2} \Rightarrow $ It has bent shape

-The molecular shape of $S{O_2}$ is V-shaped or bent shape.

-$Si{O_2}$ , silicon atom shows tetrahedral coordination, with four oxygen atoms surrounding a central silicon atom

Hence option A is correct .
Additional information :
Though VSEPR theory is able to explain the shapes of simple molecules but it is unable to predict the shape in a number of cases . Thus , it had a limited application only . Moreover , talking of the direction of electron pairs does not seem to be very rational . Thus , Lewis' approach as well as VSEPR theory had a number of limitations .
To explain the above limitations , two important theories called modern theories of covalent bond formation have been put forward , that is the valence bond theory and the molecular orbital theory .
Note:
While predicting the geometry of molecules containing double bonds , it should be kept in mind that the double bond is considered one electron pair . For example , in case of ozone the central oxygen atom is considered to have two bond pairs and one lone pair and has a bent shape structure .
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




