The minimum energy required to overcome the attractive forces between an electron and the surface of $Ag$ metal is $552 \times {10^{ - 19}}J$. What will be the maximum kinetic energy of electron ejected out from $Ag$ which is being exposed to UV light of $\lambda = 360A^\circ $
Answer
600k+ views
Hint: When an electron attempts to escape from a metal surface it leaves a positive picture behind on the metal surface. The negative electron switches back to the metal surface due to the attractiveness of this positive image, so it does not permanently leave the metal crystal. But an electron requires adequate energy supplied from outside, typically from external light sources, to overcome the attraction factor. The total energy absorbed by the electron will be the net amount of both energy required to overcome the attractive forces and kinetic energy of the electron.
Formula used:
$E = \dfrac{{hC}}{\lambda }$ where,
$E = $energy absorbed from the light
$h = 6.626 \times {10^{ - 34}}Js$ which is planck's constant
$C = 3 \times {10^8}m/s$ which is the velocity of light
Complete step by step answer:
The given values are,
Energy required to overcome the attractive force $({E_1}) = 552 \times {10^{ - 19}}J$
The wavelength of light $\lambda = 360A^\circ $ which is equal to $360 \times {10^{ - 10}}m$
By Substituting the values in equation $E = \dfrac{{hC}}{\lambda }$
$E = \dfrac{{6.626 \times {{10}^{ - 34}}J \times 3 \times {{10}^8}m/s}}{{360 \times {{10}^{ - 10}}m}}$
$ = 5.522 \times {10^{ - 18}}J$
We know that here $E$ is the ${E_{total}}$ which is the sum of both energy to overcome attractive forces $({E_1})$ and kinetic energy of electrons $({E_k})$
So ${E_{total}} = {E_1} + {E_k}$
Therefore, ${E_k} = {E_{total}} - {E_1}$
$ = (5.52 \times {10^{ - 18}}) - (552 \times {10^{ - 19}})$
$ = - 4.968 \times {10^{ - 17}}J$
This is the required maximum kinetic energy of an electron.
Additional information:
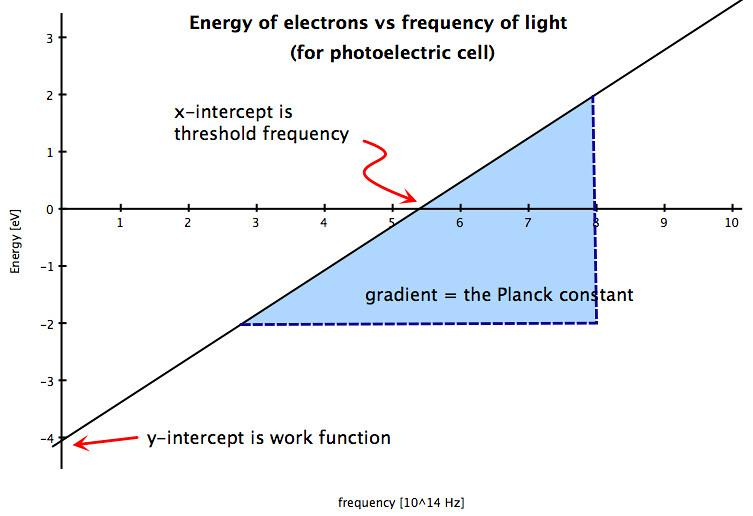
The minimum energy for an electron to overcome the attractive forces and to escape from the metal surface by the striking of a photon is called its work function and is denoted by the symbol $\phi $.The minimum frequency for this emission is called threshold frequency. Energy in always directly proportional to frequency
$E = h\nu $ where $E$ is the energy , $h$ is the planck's constant and $\nu $ is the frequency. The relation is shown by the following graph.
Note:
The energy to overcome the surface is given in $J$, so while doing the calculations take Planck's constant value also in joule. And wavelengths should need to convert into meters since we are taking the value of velocity of light in $m/s$.
Formula used:
$E = \dfrac{{hC}}{\lambda }$ where,
$E = $energy absorbed from the light
$h = 6.626 \times {10^{ - 34}}Js$ which is planck's constant
$C = 3 \times {10^8}m/s$ which is the velocity of light
Complete step by step answer:
The given values are,
Energy required to overcome the attractive force $({E_1}) = 552 \times {10^{ - 19}}J$
The wavelength of light $\lambda = 360A^\circ $ which is equal to $360 \times {10^{ - 10}}m$
By Substituting the values in equation $E = \dfrac{{hC}}{\lambda }$
$E = \dfrac{{6.626 \times {{10}^{ - 34}}J \times 3 \times {{10}^8}m/s}}{{360 \times {{10}^{ - 10}}m}}$
$ = 5.522 \times {10^{ - 18}}J$
We know that here $E$ is the ${E_{total}}$ which is the sum of both energy to overcome attractive forces $({E_1})$ and kinetic energy of electrons $({E_k})$
So ${E_{total}} = {E_1} + {E_k}$
Therefore, ${E_k} = {E_{total}} - {E_1}$
$ = (5.52 \times {10^{ - 18}}) - (552 \times {10^{ - 19}})$
$ = - 4.968 \times {10^{ - 17}}J$
This is the required maximum kinetic energy of an electron.
Additional information:
The minimum energy for an electron to overcome the attractive forces and to escape from the metal surface by the striking of a photon is called its work function and is denoted by the symbol $\phi $.The minimum frequency for this emission is called threshold frequency. Energy in always directly proportional to frequency
$E = h\nu $ where $E$ is the energy , $h$ is the planck's constant and $\nu $ is the frequency. The relation is shown by the following graph.

Note:
The energy to overcome the surface is given in $J$, so while doing the calculations take Planck's constant value also in joule. And wavelengths should need to convert into meters since we are taking the value of velocity of light in $m/s$.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




