The increasing order of $\text{ p}{{\text{K}}_{\text{b}}}\text{ }$ of the following compound is:
A)
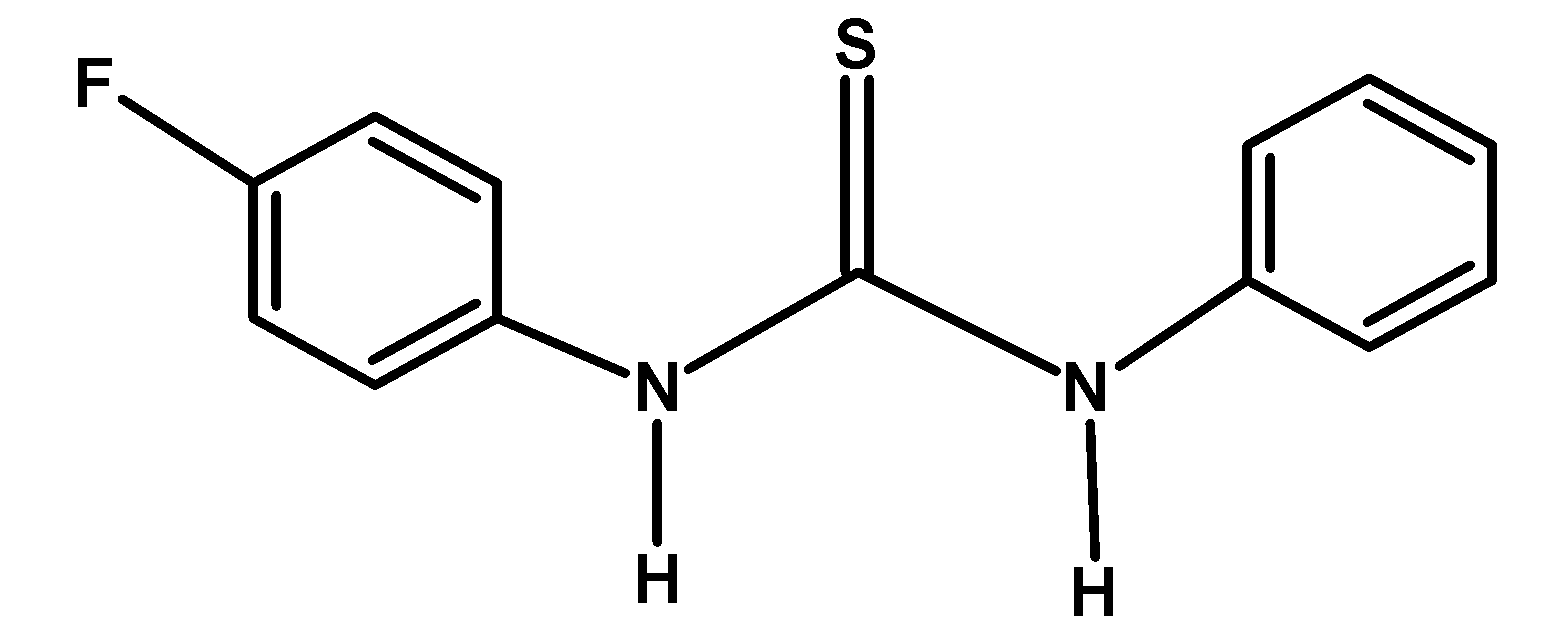
B)

C)
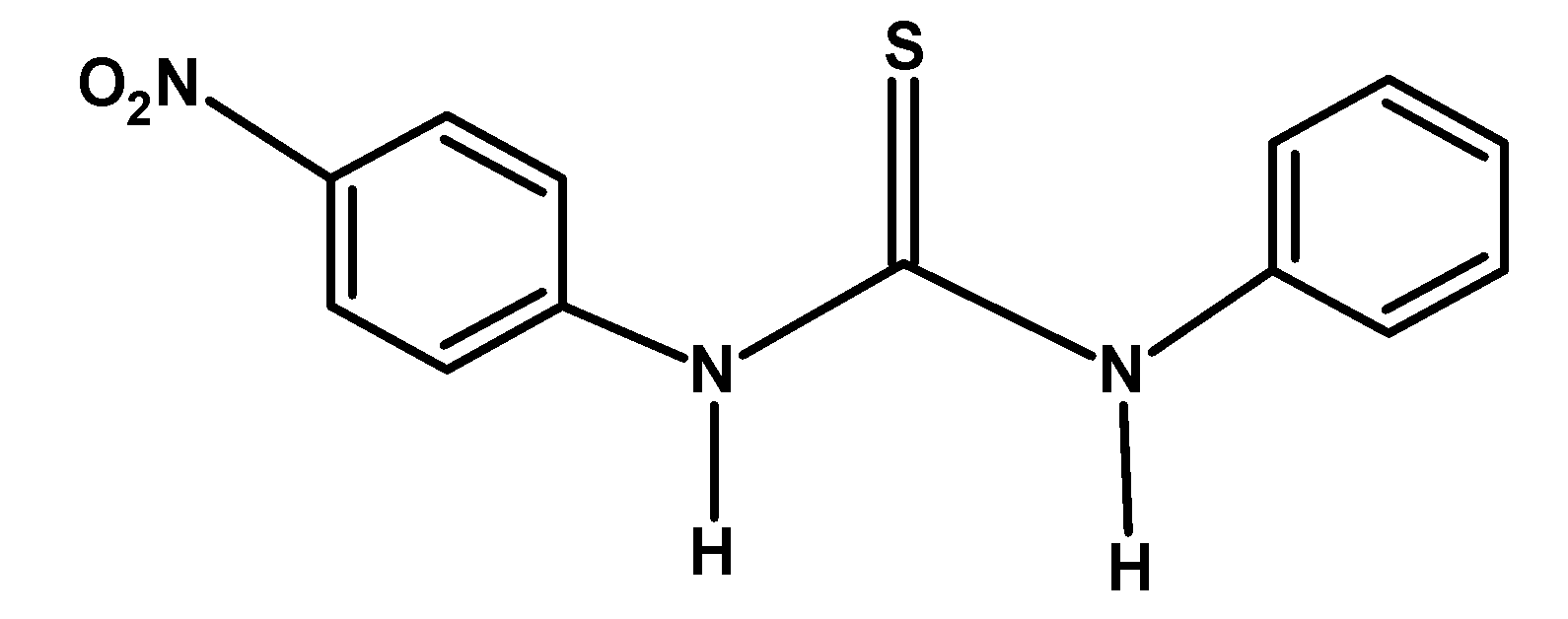
D)
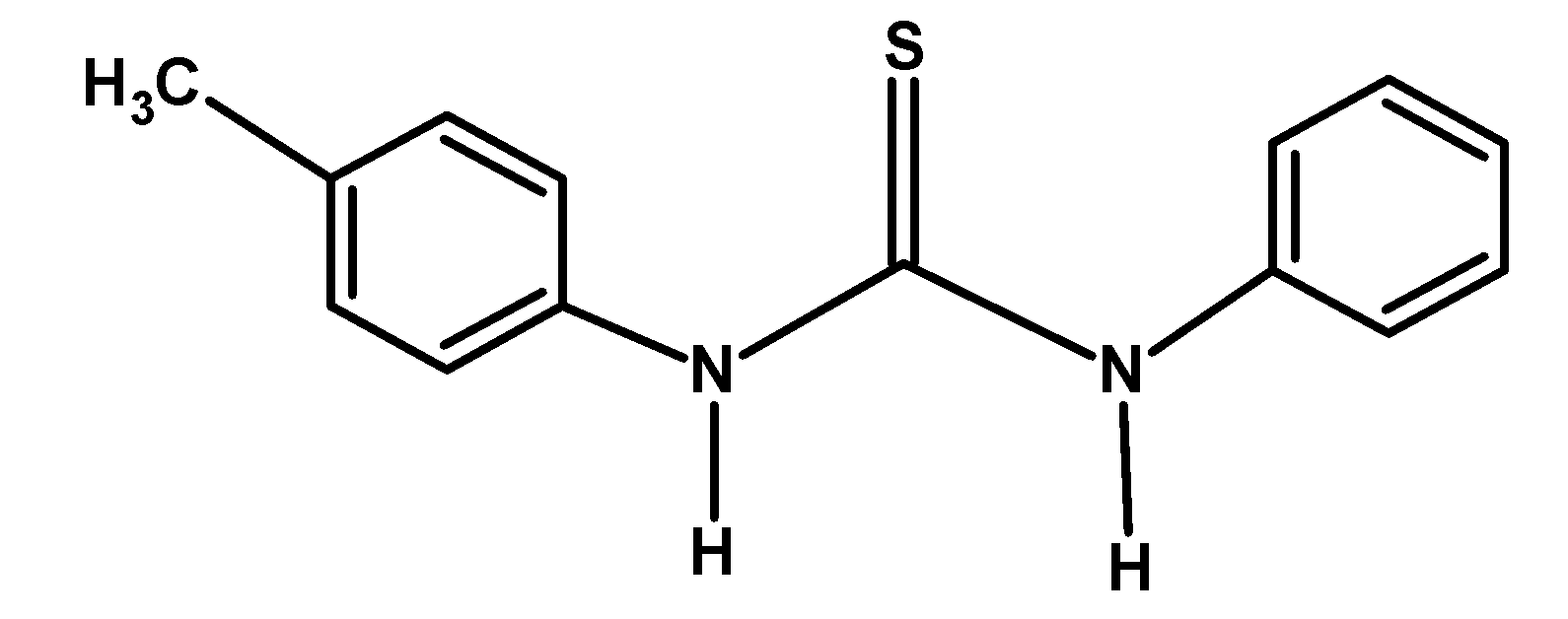
A) $\text{ A }<\text{ C }<\text{ D }<\text{ B }$
B) $\text{ B }<\text{ D }<\text{ A }<\text{ C }$
C) $\text{ C }<\text{ A }<\text{ D }<\text{ B }$
D) $\text{ B }<\text{ D }<\text{ C }<\text{ A }$
| A) | 
|
| B) | 
|
| C) | 
|
| D) | 
|
Answer
594k+ views
Hint:. The basicity is the measure of the extent of the base to accept the proton or donate the electron pair. The basicity is expressed as a dissociation constant of the base $\text{ }{{\text{K}}_{\text{b}}}$ . The basicity of an organic compound depends on the substituents. The electron releasing group increases the basicity of the base while the electron-withdrawing group decreases the basicity of the compound.
Complete step by step answer:
The basicity is determined by the $\text{ }{{\text{K}}_{\text{b}}}$ value of a base. the base tends to abstract a proton or donate an electron pair. The value of the $\text{ }{{\text{K}}_{\text{b}}}$ is equilibrium constant for the dissociation of the base.
The basicity depends on the substituents present in the compound.
The electron-donating group likes $-\text{C}{{\text{H}}_{\text{3}}}\text{ }$ and $-\text{OC}{{\text{H}}_{\text{3}}}\text{ }$releases the electron density towards the nitrogen of the compound. This stabilized the cation formed after donating an electron pair or accepting a proton. Thus from given compounds, compound B) and D) have high basicity.
Among $-\text{C}{{\text{H}}_{\text{3}}}\text{ }$ and $-\text{OC}{{\text{H}}_{\text{3}}}\text{ }$ groups, the methyl group donates its electron via the hyperconjugation and inductive effect. However, the methoxy $-\text{OC}{{\text{H}}_{\text{3}}}\text{ }$group donates the electron via the resonance. Thus compound B) has more basicity compared to D).
The electron-withdrawing group like $-\text{F }$ and $-\text{N}{{\text{O}}_{\text{2}}}\text{ }$ withdraws the electron density towards itself from the nitrogen of the compound. This destabilized the cation formed after donating an electron pair or accepting a proton. Thus from given compounds, compound A) and C) have low basicity.
Among $-\text{F }$ and $-\text{N}{{\text{O}}_{\text{2}}}\text{ }$ groups, the Florine group withdraws its electron via the inductive effect. However, the nitro$-\text{N}{{\text{O}}_{\text{2}}}\text{ }$ group withdraws the electron via the resonance. Thus compound A) has more basicity compared to C).
Thus the order of basicity for the compounds is given as,
$\text{ B }>\text{ D }>\text{ A }>\text{ C }$
The $\text{ }{{\text{K}}_{\text{b}}}$ is related to $\text{ p}{{\text{K}}_{\text{b}}}\text{ }$ is given as,’
$\text{ p}{{\text{K}}_{\text{b}}}\text{ }=\text{ }-\log {{\text{K}}_{\text{b}}}\text{ }$
Thus, the order of increasing $\text{ p}{{\text{K}}_{\text{b}}}\text{ }$ is given as,
$\text{ B }<\text{ D }<\text{ A }<\text{ C }$
So, the correct answer is “Option C”.
Note: Note that, we can summarise the basicity and $\text{ p}{{\text{K}}_{\text{b}}}\text{ }$as basicity is directly proportional to the resonance effect of electron releasing group and hyperconjugation effect of electron-donating group. However, the basicity is inversely related to the resonance effect of the electron-withdrawing group and hyperconjugation effect. Always remember that the resonance effect is the permanent effect but hyperconjugation is a temporary effect, thus resonance makes it more stable.
$\text{ Basicity }\propto \text{ +R }\propto \text{ }\dfrac{1}{-\text{R}}\propto \text{ +H }\propto \text{ }\dfrac{1}{-\text{H}}\text{ }$
Complete step by step answer:
The basicity is determined by the $\text{ }{{\text{K}}_{\text{b}}}$ value of a base. the base tends to abstract a proton or donate an electron pair. The value of the $\text{ }{{\text{K}}_{\text{b}}}$ is equilibrium constant for the dissociation of the base.
The basicity depends on the substituents present in the compound.
The electron-donating group likes $-\text{C}{{\text{H}}_{\text{3}}}\text{ }$ and $-\text{OC}{{\text{H}}_{\text{3}}}\text{ }$releases the electron density towards the nitrogen of the compound. This stabilized the cation formed after donating an electron pair or accepting a proton. Thus from given compounds, compound B) and D) have high basicity.
Among $-\text{C}{{\text{H}}_{\text{3}}}\text{ }$ and $-\text{OC}{{\text{H}}_{\text{3}}}\text{ }$ groups, the methyl group donates its electron via the hyperconjugation and inductive effect. However, the methoxy $-\text{OC}{{\text{H}}_{\text{3}}}\text{ }$group donates the electron via the resonance. Thus compound B) has more basicity compared to D).
The electron-withdrawing group like $-\text{F }$ and $-\text{N}{{\text{O}}_{\text{2}}}\text{ }$ withdraws the electron density towards itself from the nitrogen of the compound. This destabilized the cation formed after donating an electron pair or accepting a proton. Thus from given compounds, compound A) and C) have low basicity.
Among $-\text{F }$ and $-\text{N}{{\text{O}}_{\text{2}}}\text{ }$ groups, the Florine group withdraws its electron via the inductive effect. However, the nitro$-\text{N}{{\text{O}}_{\text{2}}}\text{ }$ group withdraws the electron via the resonance. Thus compound A) has more basicity compared to C).
Thus the order of basicity for the compounds is given as,
$\text{ B }>\text{ D }>\text{ A }>\text{ C }$
The $\text{ }{{\text{K}}_{\text{b}}}$ is related to $\text{ p}{{\text{K}}_{\text{b}}}\text{ }$ is given as,’
$\text{ p}{{\text{K}}_{\text{b}}}\text{ }=\text{ }-\log {{\text{K}}_{\text{b}}}\text{ }$
Thus, the order of increasing $\text{ p}{{\text{K}}_{\text{b}}}\text{ }$ is given as,
$\text{ B }<\text{ D }<\text{ A }<\text{ C }$
So, the correct answer is “Option C”.
Note: Note that, we can summarise the basicity and $\text{ p}{{\text{K}}_{\text{b}}}\text{ }$as basicity is directly proportional to the resonance effect of electron releasing group and hyperconjugation effect of electron-donating group. However, the basicity is inversely related to the resonance effect of the electron-withdrawing group and hyperconjugation effect. Always remember that the resonance effect is the permanent effect but hyperconjugation is a temporary effect, thus resonance makes it more stable.
$\text{ Basicity }\propto \text{ +R }\propto \text{ }\dfrac{1}{-\text{R}}\propto \text{ +H }\propto \text{ }\dfrac{1}{-\text{H}}\text{ }$
Recently Updated Pages
Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Explain the structure of megasporangium class 12 biology CBSE

What are the major means of transport Explain each class 12 social science CBSE

How many chromosomes are found in human ovum a 46 b class 12 biology CBSE

The diagram of the section of a maize grain is given class 12 biology CBSE




