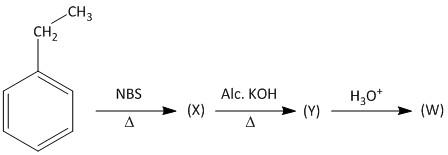
The end product ‘W’ in the following sequence of reaction is:
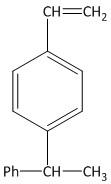
(A)
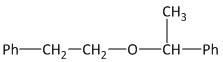
(B)
(C) $Ph - C{H_2} - O - C{H_2} - C{H_2} - Ph$
(D)

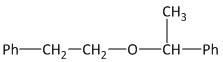


Answer
570.3k+ views
Hint: The \[NBS\] is a reagent used for benzylic bromination. The middle step is an elimination reaction. The final step is a hydration reaction of alkene.
Complete step by step answer:
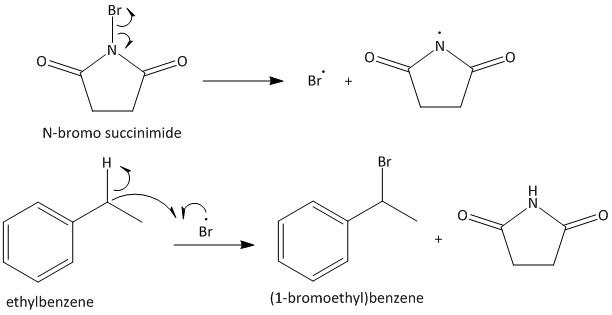
The given starting material for the reaction sequence is ethyl benzene. It is an aromatic hydrocarbon which undergoes benzylic bromination upon heating with \[NBS\]. \[NBS\] is a brominating agent which contains bromine radicals. The bromine radical reacts with the hydrocarbon to produce more stabilized benzyl bromide. Thus the product of the reaction is (1-bromoethyl)benzene. The reaction is shown as
Step I:
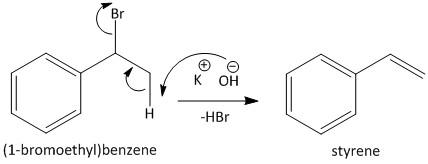
In the second step the alcoholic \[KOH\] is used to abstract the hydrogen from the methyl group of the benzyl bromide. The abstraction of the hydrogen from the compound results in dehydrobromination and produces styrene. The reaction is shown as
Step II:
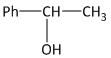
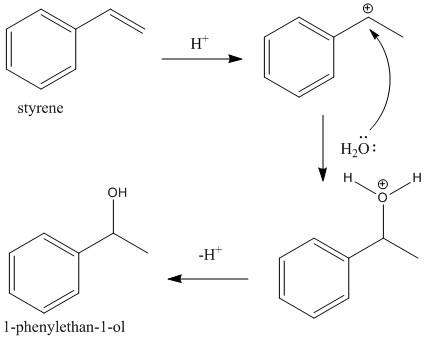
The third and the final step is the hydration of styrene molecules in presence of aqueous acid. At first the acid protonated the double bond of the styrene molecule to produce a more stable \[{2^o}\]carbocation (secondary). This then undergoes addition of water molecules to generate \[1\]-phenylethan-\[1\]-ol. The reaction is shown as
Step III:
Hence the end product ‘W’ of the given sequence of reaction is \[1\]-phenylethan-\[1\]-ol, i.e. option B is the correct answer.
So, the correct answer is Option B.
Note: The reaction conditions and the abbreviations are to be kept in mind. The alc. is used for alcoholic \[KOH\] and aq. is used for aqueous \[KOH\]. Alc. \[KOH\] will result in elimination of the product but the aqueous \[KOH\] is used for hydroxylation i.e. addition of \[OH\].
Complete step by step answer:
The given starting material for the reaction sequence is ethyl benzene. It is an aromatic hydrocarbon which undergoes benzylic bromination upon heating with \[NBS\]. \[NBS\] is a brominating agent which contains bromine radicals. The bromine radical reacts with the hydrocarbon to produce more stabilized benzyl bromide. Thus the product of the reaction is (1-bromoethyl)benzene. The reaction is shown as
Step I:

In the second step the alcoholic \[KOH\] is used to abstract the hydrogen from the methyl group of the benzyl bromide. The abstraction of the hydrogen from the compound results in dehydrobromination and produces styrene. The reaction is shown as
Step II:

The third and the final step is the hydration of styrene molecules in presence of aqueous acid. At first the acid protonated the double bond of the styrene molecule to produce a more stable \[{2^o}\]carbocation (secondary). This then undergoes addition of water molecules to generate \[1\]-phenylethan-\[1\]-ol. The reaction is shown as
Step III:

Hence the end product ‘W’ of the given sequence of reaction is \[1\]-phenylethan-\[1\]-ol, i.e. option B is the correct answer.
So, the correct answer is Option B.
Note: The reaction conditions and the abbreviations are to be kept in mind. The alc. is used for alcoholic \[KOH\] and aq. is used for aqueous \[KOH\]. Alc. \[KOH\] will result in elimination of the product but the aqueous \[KOH\] is used for hydroxylation i.e. addition of \[OH\].
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




