The artificial sweetener containing chlorine that has the appearance and taste as that of sugar and is stable at cooking temperature is-
A) Aspartame
B) Saccharin
C) Sucralose
D) Alitame
Answer
594.9k+ views
Hint: Alitame and Sucralose are the only artificial sweeteners that are stable at cooking temperatures.
Complete step by step answer:
The artificial sweetener containing chlorine that has the appearance and taste as that of sugar and is stable at cooking temperature is Sucralose. Its chemical formula is\[{{\text{C}}_{{\text{12}}}}{{\text{H}}_{{\text{19}}}}{\text{C}}{{\text{l}}_{\text{3}}}{{\text{O}}_{\text{8}}}\] .Its structure is-
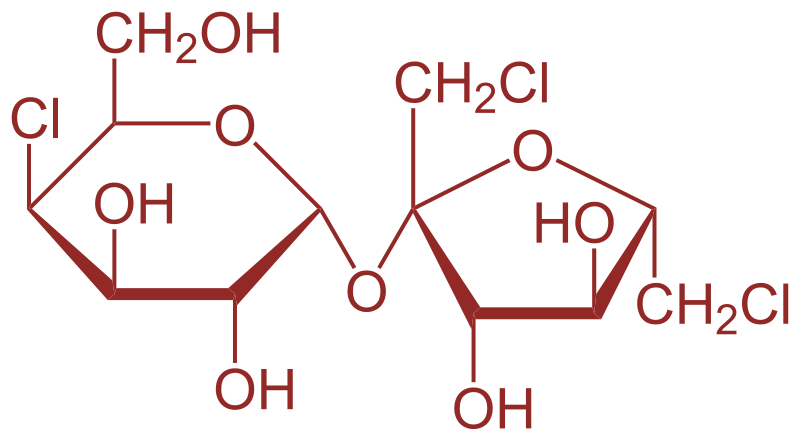
It is derived from sugar through a chemical process in which $3$ hydroxyl groups are replaced by $3$ chlorine atoms on sugar molecules which produces a sweetener which has no calories and is $600$ times sweeter than sucrose. In Sucralose, ${\text{4 - chloro - 4 - deoxy - alpha - D - galactopyranose}}$ and ${\text{1,6 - dichloro - 1,6 - dideoxy - beta - D - fructofuranose}}$ are the units which are linked by glycosidic bond.
The properties of Sucralose are-
-It is a disaccharide derivative and since it has chlorine in its formula it is also known as organo-chlorine compound.
-It is a white crystalline powder and has a sweet taste.
-It has a melting point of ${130^ \circ }{\text{C}}$.
-It is soluble in water, methanol and ethanol but only slightly soluble in ethyl acetate.
-It also plays a role as an environmental contaminant.
Hence, the correct answer is ‘C’.
Note:
Aspartame and saccharin decompose at cooking temperature hence they are not used in baked foods. Alitame does not contain chlorine so only Sucralose is the possible answer. The uses of Sucralose are –
-It is used as a food additive and flavor enhancer.
-It is also a xenobiotic agent.
Complete step by step answer:
The artificial sweetener containing chlorine that has the appearance and taste as that of sugar and is stable at cooking temperature is Sucralose. Its chemical formula is\[{{\text{C}}_{{\text{12}}}}{{\text{H}}_{{\text{19}}}}{\text{C}}{{\text{l}}_{\text{3}}}{{\text{O}}_{\text{8}}}\] .Its structure is-

It is derived from sugar through a chemical process in which $3$ hydroxyl groups are replaced by $3$ chlorine atoms on sugar molecules which produces a sweetener which has no calories and is $600$ times sweeter than sucrose. In Sucralose, ${\text{4 - chloro - 4 - deoxy - alpha - D - galactopyranose}}$ and ${\text{1,6 - dichloro - 1,6 - dideoxy - beta - D - fructofuranose}}$ are the units which are linked by glycosidic bond.
The properties of Sucralose are-
-It is a disaccharide derivative and since it has chlorine in its formula it is also known as organo-chlorine compound.
-It is a white crystalline powder and has a sweet taste.
-It has a melting point of ${130^ \circ }{\text{C}}$.
-It is soluble in water, methanol and ethanol but only slightly soluble in ethyl acetate.
-It also plays a role as an environmental contaminant.
Hence, the correct answer is ‘C’.
Note:
Aspartame and saccharin decompose at cooking temperature hence they are not used in baked foods. Alitame does not contain chlorine so only Sucralose is the possible answer. The uses of Sucralose are –
-It is used as a food additive and flavor enhancer.
-It is also a xenobiotic agent.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

What are the major means of transport Explain each class 12 social science CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




