The acid present in the body of red ant is ______.
Answer
632.4k+ views
Hint: An ant is called ‘Formica’ in Latin. The acid found in red ants is the simplest carboxylic acid.
Complete step by step answer:
The acid found in the body of red ant is formic acid, named after the Latin name of an ant ‘Formica’. The IUPAC name of formic acid is methanoic acid. Methanoic acid (HCOOH) is the simplest carboxylic acid. The irritating itching sensation after an ant’s bite is thus due to formic acid. Formic acid is dangerous to the human body at high concentrations, but very useful at low concentrations. It is used as a food preservative due to its antibacterial nature. It is also used in pesticides and cosmetic additives. It is also used to help a variety of industrial processes to occur. Methanoic acid consists of a carbon doubly bonded to an oxygen and a single bond joining it to a hydroxyl (OH) group. The structure of formic acid is:
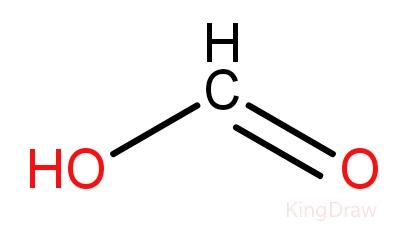
Additional information: Our bodies also make small amounts of formic acid from the methanol we intake. Some of the methanol is also produced in our body from aspartame. Our body converts aspartame into aspartic acid, methanol and phenylalanine. The methanol is therefore converted into formic acid.
Note: One can think formic acid to be very corrosive to skin. But formic acid is a mild naturally occurring acid. In small amounts, it is in fact very beneficial to the human body.
Complete step by step answer:
The acid found in the body of red ant is formic acid, named after the Latin name of an ant ‘Formica’. The IUPAC name of formic acid is methanoic acid. Methanoic acid (HCOOH) is the simplest carboxylic acid. The irritating itching sensation after an ant’s bite is thus due to formic acid. Formic acid is dangerous to the human body at high concentrations, but very useful at low concentrations. It is used as a food preservative due to its antibacterial nature. It is also used in pesticides and cosmetic additives. It is also used to help a variety of industrial processes to occur. Methanoic acid consists of a carbon doubly bonded to an oxygen and a single bond joining it to a hydroxyl (OH) group. The structure of formic acid is:

Additional information: Our bodies also make small amounts of formic acid from the methanol we intake. Some of the methanol is also produced in our body from aspartame. Our body converts aspartame into aspartic acid, methanol and phenylalanine. The methanol is therefore converted into formic acid.
Note: One can think formic acid to be very corrosive to skin. But formic acid is a mild naturally occurring acid. In small amounts, it is in fact very beneficial to the human body.
Recently Updated Pages
Master Class 10 Computer Science: Engaging Questions & Answers for Success

Master Class 10 General Knowledge: Engaging Questions & Answers for Success

Master Class 10 English: Engaging Questions & Answers for Success

Master Class 10 Social Science: Engaging Questions & Answers for Success

Master Class 10 Maths: Engaging Questions & Answers for Success

Master Class 10 Science: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




