Reaction of a carbonyl compound with one of the following reagents involves nucleophilic addition followed by elimination of water. The reagent is:
(A) Hydrocyanic acid
(B) Sodium hydrogen sulphite
(C) A Grignard reagent
(D) Hydrazine in presence of feebly acidic solution
Answer
601.2k+ views
Hint: The reagent forms imines as a final product which does nucleophilic attack on carbonyl carbon followed by the loss of water. The reagent has a nucleophilic nitrogen atom in its structure.
Complete step by step solution:
We will take a look at all the given reagents in order to find the answer.
- Hydrocyanic acid or HCN has cyanide ion as a nucleophile. It can attack the carbonyl group of carbon and form cyanohydrins. The cyanohydrins are stable enough that they do not lose a molecule of water. Thus, it is not the correct answer.
- Sodium hydrogen sulphite or $NaHS{O_3}$ is an ionic compound and does not give any nucleophilic addition reaction with carbonyl groups.
- A Grignard reagent can attack the carbonyl group of the carbon via a nucleophile but the reaction does not involve loss of water molecule.
- When a carbonyl compound is allowed to react with hydrazine in a feebly acidic medium, the nucleophilic nitrogen atom attacks the electrophilic carbon atom of the carbonyl group. Then, Water molecule is lost in the presence of a weak acidic medium and an imine is formed as a final product. Here, a weak acidic medium helps the hydroxyl group to leave. The reaction can be shown as below.
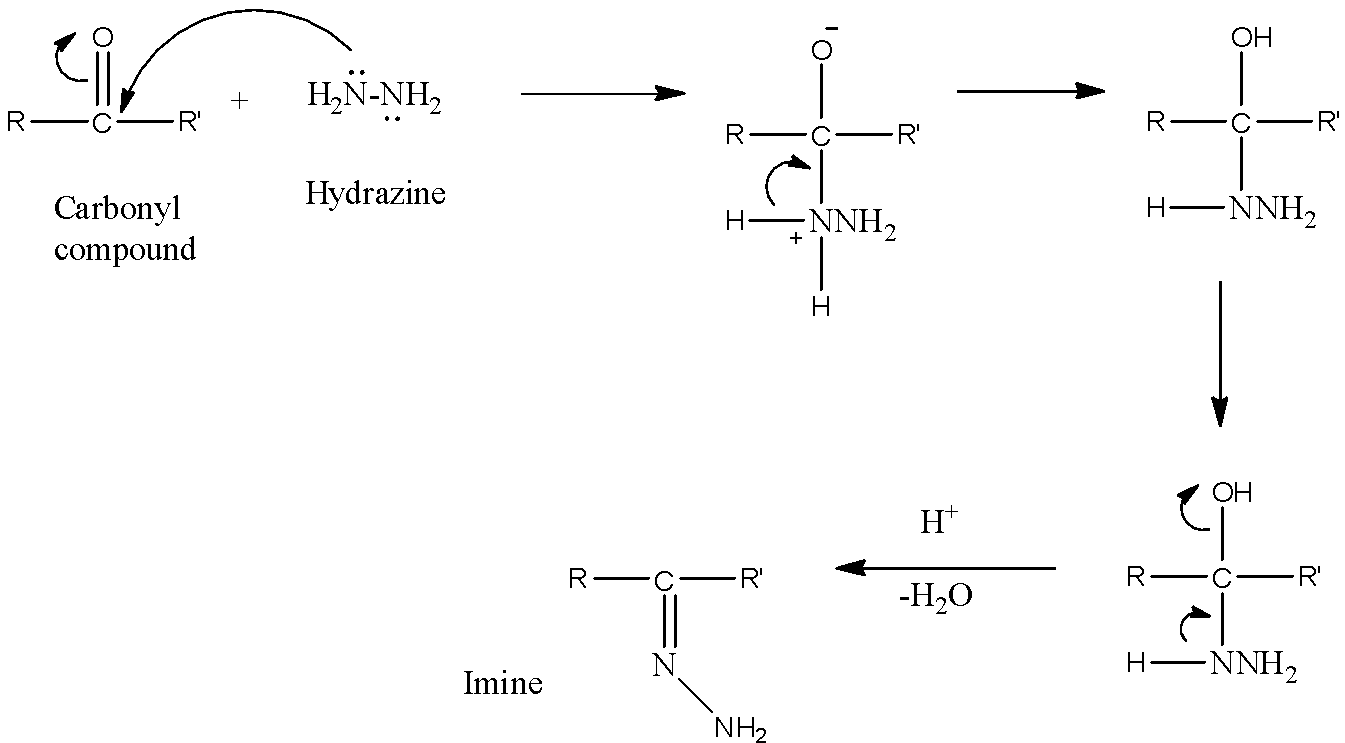
So, we can conclude that when a carbonyl compound reacts with hydrazine in a feebly acidic solution, then nucleophilic attack is involved and imine is formed.
Thus, we can say that the correct option is (D).
Note: Note that the $\alpha $-hydroxy amines are not stable and so that they always form imines upon losing water molecules. Sodium hydrogen sulphite is a weak base and it only reacts with strongly acidic compounds.
Complete step by step solution:
We will take a look at all the given reagents in order to find the answer.
- Hydrocyanic acid or HCN has cyanide ion as a nucleophile. It can attack the carbonyl group of carbon and form cyanohydrins. The cyanohydrins are stable enough that they do not lose a molecule of water. Thus, it is not the correct answer.
- Sodium hydrogen sulphite or $NaHS{O_3}$ is an ionic compound and does not give any nucleophilic addition reaction with carbonyl groups.
- A Grignard reagent can attack the carbonyl group of the carbon via a nucleophile but the reaction does not involve loss of water molecule.
- When a carbonyl compound is allowed to react with hydrazine in a feebly acidic medium, the nucleophilic nitrogen atom attacks the electrophilic carbon atom of the carbonyl group. Then, Water molecule is lost in the presence of a weak acidic medium and an imine is formed as a final product. Here, a weak acidic medium helps the hydroxyl group to leave. The reaction can be shown as below.

So, we can conclude that when a carbonyl compound reacts with hydrazine in a feebly acidic solution, then nucleophilic attack is involved and imine is formed.
Thus, we can say that the correct option is (D).
Note: Note that the $\alpha $-hydroxy amines are not stable and so that they always form imines upon losing water molecules. Sodium hydrogen sulphite is a weak base and it only reacts with strongly acidic compounds.
Recently Updated Pages
Master Class 10 Computer Science: Engaging Questions & Answers for Success

Master Class 10 General Knowledge: Engaging Questions & Answers for Success

Master Class 10 English: Engaging Questions & Answers for Success

Master Class 10 Social Science: Engaging Questions & Answers for Success

Master Class 10 Maths: Engaging Questions & Answers for Success

Master Class 10 Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




