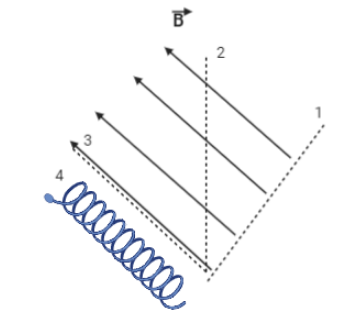
Radiation coming from a radioactive substance is passed through a cloud-chamber in which a strong constant magnetic field is switched on in the direction shown in figure. Figure shows a path followed by a few particles coming from radioactive substances (shown by dotted lines). Neglect the interaction between different particles. Tick the correct statements.
This question has multiple correct options.
A. Path 1 can be followed by $\gamma $ -radiation only
B. Path 3 can be followed by$\gamma $ -radiation only
C. Path 4 can be followed by $\alpha $ radiation only
D. Path 4 can be followed by $\beta $ radiation.

Answer
585.9k+ views
Hint:The $\alpha $-particles carry positive charge, $\beta $-particles carry negative charge and $\gamma $-particles are uncharged particles. The uncharged particles are not deflected by magnetic fields and both alpha and beta particles are deflected by both the electric and magnetic fields.
Complete answer:
A radioactive substance is emitting radiations and it is passed through a cloud-chamber in which a strong intensity of magnetic field is switched on in the direction shown by the four arrows. A cloud chamber is a device which is used to detect ionizing particles and it is also used to determine the trajectories i.e., the path followed by the different particles when it passes through the chamber. The three dotted lines and spiral shape shows the path followed by the few particles.
$\alpha $ , $\beta $ and $\gamma $ are the types of radiations. Alpha $\left( \alpha \right)$ particles are positively charged, beta $\left( \beta \right)$ particles are negatively charged and gamma $\left( \gamma \right)$ particles do not carry any charge. The alpha and beta particles are deflected by both the electric and magnetic fields as they carry charge. The deflection of alpha particles is less as compared to that of beta particles because alpha particles are more massive than beta particles and deflection of beta particles is in a direction opposite to that of alpha particle since both of them carry opposite charge.
Hence, path 1 is represented by $\gamma $ -radiation only as it is not deflected by the magnetic field i.e., it travels in a straight line. Path 4 can be followed by $\beta $-radiation because it is deflected by the magnetic field. Option C is wrong because path 4 can be followed both by $\alpha $ and $\beta $ radiations. So, the word only is not correct in option C. The $\beta $ -particle can be deflected in a straight line or in a spiral shape and it depends on the velocity of the particle.
Therefore, options A and D are correct.
Note: Kindly remember that path 1 can only be followed by $\gamma $-particles as it does not carry any charge. Thus, it would not be deflected by a magnetic field. Path 4 can be followed by both $\alpha $ and β particles because they carry charge and spiral shape depends on the speed of the particles.
Complete answer:
A radioactive substance is emitting radiations and it is passed through a cloud-chamber in which a strong intensity of magnetic field is switched on in the direction shown by the four arrows. A cloud chamber is a device which is used to detect ionizing particles and it is also used to determine the trajectories i.e., the path followed by the different particles when it passes through the chamber. The three dotted lines and spiral shape shows the path followed by the few particles.
$\alpha $ , $\beta $ and $\gamma $ are the types of radiations. Alpha $\left( \alpha \right)$ particles are positively charged, beta $\left( \beta \right)$ particles are negatively charged and gamma $\left( \gamma \right)$ particles do not carry any charge. The alpha and beta particles are deflected by both the electric and magnetic fields as they carry charge. The deflection of alpha particles is less as compared to that of beta particles because alpha particles are more massive than beta particles and deflection of beta particles is in a direction opposite to that of alpha particle since both of them carry opposite charge.
Hence, path 1 is represented by $\gamma $ -radiation only as it is not deflected by the magnetic field i.e., it travels in a straight line. Path 4 can be followed by $\beta $-radiation because it is deflected by the magnetic field. Option C is wrong because path 4 can be followed both by $\alpha $ and $\beta $ radiations. So, the word only is not correct in option C. The $\beta $ -particle can be deflected in a straight line or in a spiral shape and it depends on the velocity of the particle.
Therefore, options A and D are correct.
Note: Kindly remember that path 1 can only be followed by $\gamma $-particles as it does not carry any charge. Thus, it would not be deflected by a magnetic field. Path 4 can be followed by both $\alpha $ and β particles because they carry charge and spiral shape depends on the speed of the particles.
Recently Updated Pages
How is Abiogenesis Theory Disproved Experimentally?

In a plane electromagnetic wave the electric field class 12 physics CBSE

A plane electromagnetic wave travels in vacuum along class 12 physics CBSE

The branch of science which deals with nature and natural class 10 physics CBSE

Understanding the Sun's Density: Exploring the Mass Density of a Hot Plasma - FAQs and Data Analysis

Where is the Centre for Environmental Education Located?

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Mention the role of cyanobacteria as a biofertiliz class 12 biology ICSE

An example of C4 plant is A Maize B Rice C Wheat D class 12 biology CBSE

Where does fertilization take place in human beings class 12 biology CBSE

Which of the following is the cheapest mode of transport class 12 social science CBSE




