How can you prepare ethyl acetate from acetic acid?
Answer
578.1k+ views
Hint: Ethyl acetate is an ester of acetic acid and ethanol, with chemical formula $[C{H_3}COOC{H_2}C{H_3}]$. It is a colourless compound with sweet and fruity odour. It is a volatile compound. It is most commonly found ester in fruits.
Acetic acid or ethanoic acid is a carboxylic acid and is a constituent of vinegar. The chemical formula of acetic acid is $[C{H_3}COOH]$.
Complete step by step answer:
As we know, ethyl acetate is an ester. Given below is the structure of ethyl acetate.
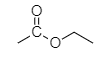
To prepare ethyl acetate from acetic acid, esterification of acetic acid should be carried out.
When acetic acid reacts with ethanol in the presence of sulphuric acid, ethyl acetate is produced through the process of esterification. Esterification is the chemical reaction between carboxylic acid and an alcohol to produce an ester.
In this chemical reaction, ${H_2}S{O_4}$ acts as a dehydrating agent and eliminates water molecules to produce ester. The process of esterification is a reversible reaction and is quite a slow process. The ${H_2}S{O_4}$ also acts as a catalyst in this reaction.
The chemical reaction for the formation of ethyl acetate from acetic acid is shown below.
$C{H_3}COOH + C{H_3}C{H_2}OH \rightleftharpoons C{H_3}COOC{H_3}C{H_2} + {H_2}O$
The reaction is carried out by heating the reactants in the presence of ${H_2}S{O_4}$.
Additional information:
Ethyl acetate is an ester and esters are extensively used in fragrance, food flavouring and in cosmetics due to its pleasant smell. Esters are also used as an organic solvent. The polymer of ester that is polyester is used in the production of plastic.
Note: The esterification reaction involves combining carboxylic acid and an alcohol in presence of ${H_2}S{O_4}$ to obtain an ester. Remember that the alcohol mentioned here should be a primary alcohol.
Esterification can also take place by reacting an acid anhydride with an alcohol or the reaction between an acid chloride with an alcohol. Here, the alcohol mentioned is the primary alcohol.
Acetic acid or ethanoic acid is a carboxylic acid and is a constituent of vinegar. The chemical formula of acetic acid is $[C{H_3}COOH]$.
Complete step by step answer:
As we know, ethyl acetate is an ester. Given below is the structure of ethyl acetate.

To prepare ethyl acetate from acetic acid, esterification of acetic acid should be carried out.
When acetic acid reacts with ethanol in the presence of sulphuric acid, ethyl acetate is produced through the process of esterification. Esterification is the chemical reaction between carboxylic acid and an alcohol to produce an ester.
In this chemical reaction, ${H_2}S{O_4}$ acts as a dehydrating agent and eliminates water molecules to produce ester. The process of esterification is a reversible reaction and is quite a slow process. The ${H_2}S{O_4}$ also acts as a catalyst in this reaction.
The chemical reaction for the formation of ethyl acetate from acetic acid is shown below.
$C{H_3}COOH + C{H_3}C{H_2}OH \rightleftharpoons C{H_3}COOC{H_3}C{H_2} + {H_2}O$
The reaction is carried out by heating the reactants in the presence of ${H_2}S{O_4}$.
Additional information:
Ethyl acetate is an ester and esters are extensively used in fragrance, food flavouring and in cosmetics due to its pleasant smell. Esters are also used as an organic solvent. The polymer of ester that is polyester is used in the production of plastic.
Note: The esterification reaction involves combining carboxylic acid and an alcohol in presence of ${H_2}S{O_4}$ to obtain an ester. Remember that the alcohol mentioned here should be a primary alcohol.
Esterification can also take place by reacting an acid anhydride with an alcohol or the reaction between an acid chloride with an alcohol. Here, the alcohol mentioned is the primary alcohol.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

What are the major means of transport Explain each class 12 social science CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




