Nitration of aniline in the strong acidic medium also gives $m - $ nitroaniline because ________.
A: In absence of substitutes, the nitro group always goes to $m - $ positions.
B: In spite of substituents, the nitro group always goes to only $m - $ positions.
C: In strong acidic medium aniline is present as anilinium ion.
D: In electrophilic substitution reactions, the amino group is Meta directive.
Answer
626.4k+ views
Hint: $m - $ nitroaniline is also called $3 - $ nitroaniline. It is a volatile substance used as a raw material in production of dyes. It is stable in neutral, basic as well as acidic medium. It is classified as not readily biodegradable.
Complete step by step answer:
In nitration of aniline nitric acid oxidizes aniline to anilinium ion. This anilinium ion is a meta directive due to which $m - $ nitroaniline is formed. ${C_6}{H_5}NH_3^ + $ is anilinium ion. In this nitrogen is already having four bonds (one with benzene ring and three with other hydrogen atoms) this means it has no lone pair. This means there will be no conjugation (shifting of negative charge) but there is positive charge on nitrogen atom this means there will be negative inductive effect due to nitrogen. Due to which position where positive charge will be farthest from nitrogen atom will be most stable also these farthest positions are para and then Meta. Due to which Meta nitroaniline is also formed.
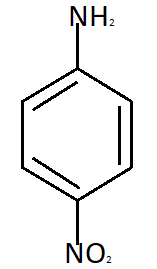
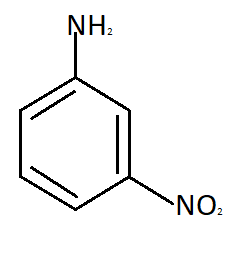
Para nitroaniline meta nitroaniline
So the reason due to which Meta nitroaniline is formed is formation of anilinium ion which imposes the inductive effect. So the correct answer is option C that is in strong acidic medium aniline is present as anilinium ion.
Note:
When an electron donating group is attached to an alkyl group then the charge is relayed through the chain it is called positive inductive effect. Similarly when an electron withdrawing group is attached negative inductive effect will take place.
Complete step by step answer:
In nitration of aniline nitric acid oxidizes aniline to anilinium ion. This anilinium ion is a meta directive due to which $m - $ nitroaniline is formed. ${C_6}{H_5}NH_3^ + $ is anilinium ion. In this nitrogen is already having four bonds (one with benzene ring and three with other hydrogen atoms) this means it has no lone pair. This means there will be no conjugation (shifting of negative charge) but there is positive charge on nitrogen atom this means there will be negative inductive effect due to nitrogen. Due to which position where positive charge will be farthest from nitrogen atom will be most stable also these farthest positions are para and then Meta. Due to which Meta nitroaniline is also formed.


Para nitroaniline meta nitroaniline
So the reason due to which Meta nitroaniline is formed is formation of anilinium ion which imposes the inductive effect. So the correct answer is option C that is in strong acidic medium aniline is present as anilinium ion.
Note:
When an electron donating group is attached to an alkyl group then the charge is relayed through the chain it is called positive inductive effect. Similarly when an electron withdrawing group is attached negative inductive effect will take place.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




