Name the compound
$C{H_3} - CH = CH - C{H_3}$
Answer
603.6k+ views
Hint: This is the example of unsaturated hydrocarbons. Unsaturated hydrocarbons are hydrocarbons that have double or triple covalent bonds between adjacent carbon atoms. The term unsaturated means more hydrogen atoms may be added to the hydrocarbons to make it saturated.
Complete step by step answer:
$C{H_3} - CH = CH - C{H_3}$
This is a But-2-ene compound. It is named so because it has a double bond on the second carbon atom .
(1) The chemical formula for butane is: ${C_4}{H_8}$ which means it is made up of four carbon atoms and eight hydrogen atoms. The $ene$ part of the name refers to an alkene. So we know that butane’s structure must include a carbon double bond.
(2) There are several different isomers or molecular structures, that this compound can form:
Additional information:
The relationships between each of these isomers are mostly constitutional, which means they have the same molecular formula but have different bond connections (Order).
Meaning to transform them, you would have to just move around the hydrogen and carbons in other words the atoms and functional groups.
Note:
These isomers of butane are made up from the same materials, they each have different properties.
With cis-beta-butylene and trans-beta-butylene the atoms are in the same order, but the polarities are not same . The $cis$ isomer is polar, with both $C{H_3}$ groups on the same side. This makes it really bulky and difficult to stack.
Complete step by step answer:
$C{H_3} - CH = CH - C{H_3}$
This is a But-2-ene compound. It is named so because it has a double bond on the second carbon atom .
(1) The chemical formula for butane is: ${C_4}{H_8}$ which means it is made up of four carbon atoms and eight hydrogen atoms. The $ene$ part of the name refers to an alkene. So we know that butane’s structure must include a carbon double bond.
(2) There are several different isomers or molecular structures, that this compound can form:
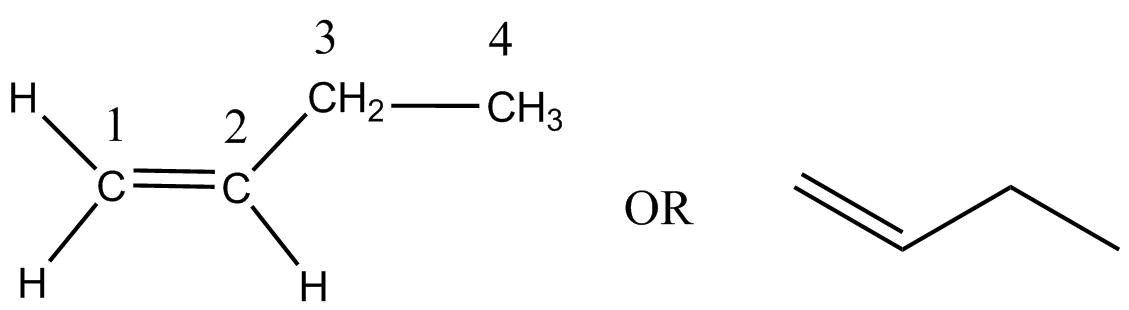
Alpha-butylene $\left( {but - 1 - ene} \right)$

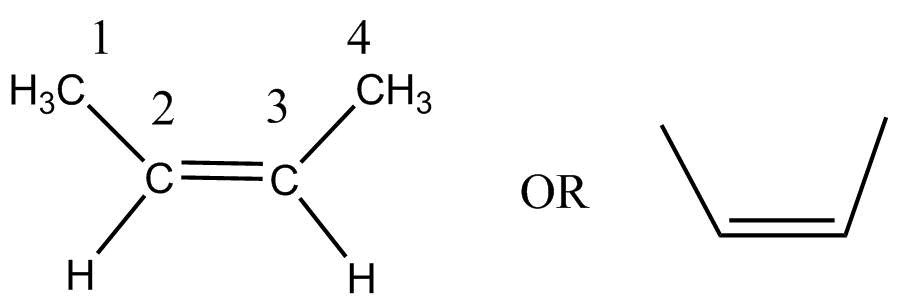
$C$ is beta-butylene- $\left( {\left( {2z} \right) - but - 2 - ene} \right)$

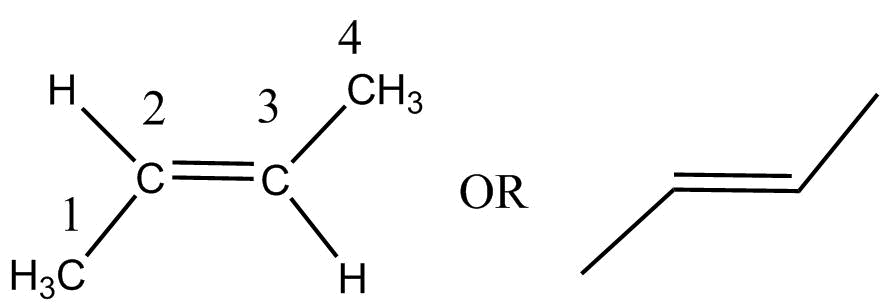
Trans-beta-butylene- $\left( {\left( {2E} \right) - but - 2ene} \right)$

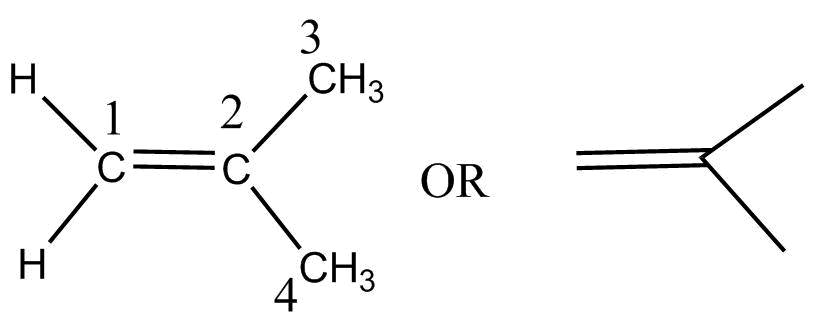
Iso butylene $\left( {2 - methylprop - 1 - ene} \right)$

Additional information:
The relationships between each of these isomers are mostly constitutional, which means they have the same molecular formula but have different bond connections (Order).
Meaning to transform them, you would have to just move around the hydrogen and carbons in other words the atoms and functional groups.
Note:
These isomers of butane are made up from the same materials, they each have different properties.
With cis-beta-butylene and trans-beta-butylene the atoms are in the same order, but the polarities are not same . The $cis$ isomer is polar, with both $C{H_3}$ groups on the same side. This makes it really bulky and difficult to stack.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




