How many more grams of \[KI\] will dissolve in $100g$ of water at ${{40}^{\circ }}C$ than at ${{20}^{\circ }}C$ ?
Answer
556.8k+ views
Hint: Here the concept of solubility works, solubility is the amount of a particular solute to dissolve in a certain amount of solvent at that temperature. We know that the solubility increases as the temperature increases. The daily life example you can take is the making of sweet syrup for sweets. Here it was asked for the extra amount that we need at these two temperatures.
Complete step by step answer:
The solubility of different solutes can be seen by using the solubility curve. In the curve we have solubility in $\left( \dfrac{g}{100}c{{m}^{3}}\,water \right)\,Vs\,\left( temperature \right)$ this graph shows how the solubility increases with the increase in temperature. So, see the plot for potassium iodide which is given below.
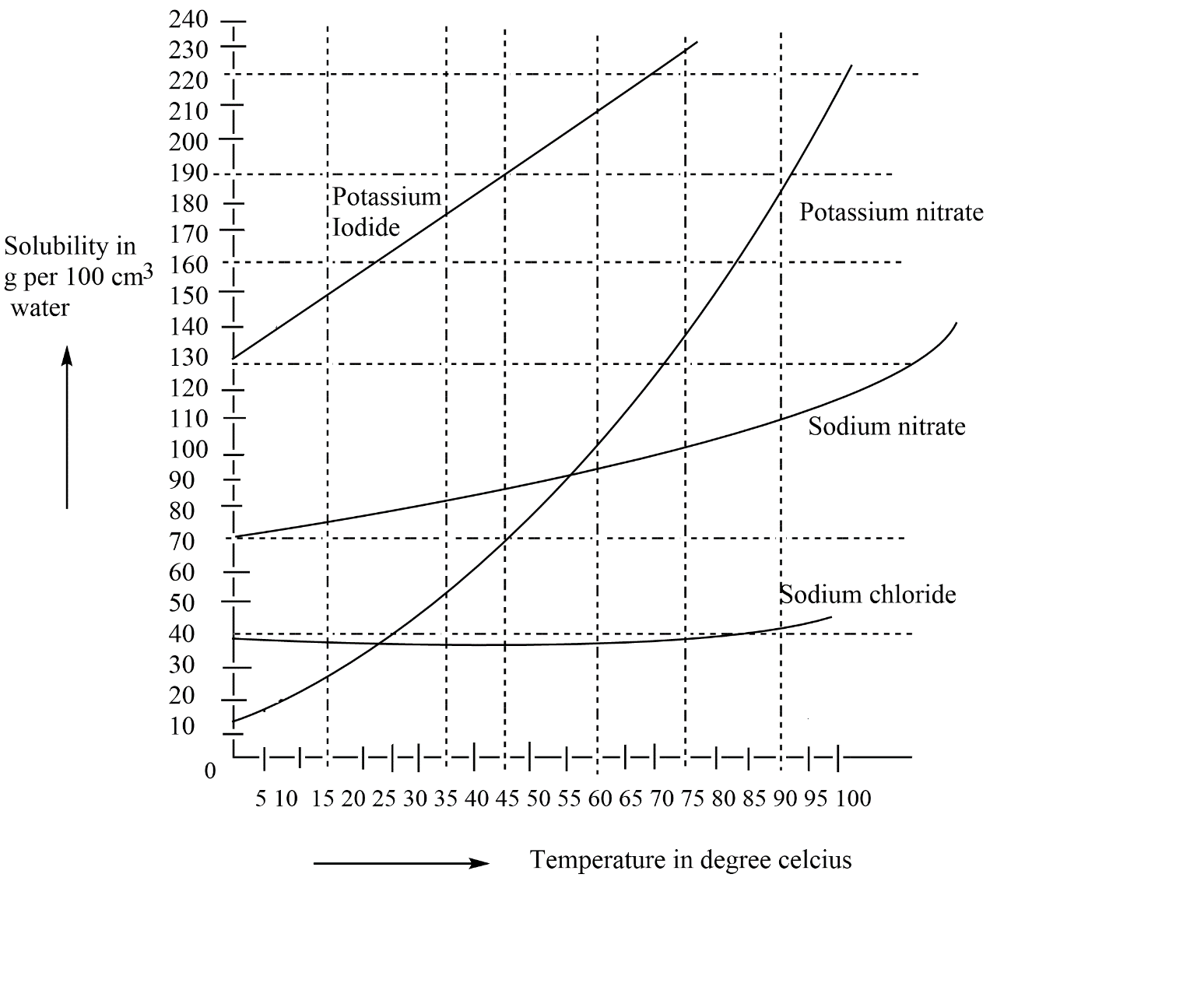
Here, as we are seeing the solubility of different salts in water and their changes with respect to temperature. So, now see for potassium iodide that at ${{40}^{\circ }}C$ the amount of potassium iodide dissolved in water is $195\,g$ you will able to see through the lines drawn so similarly of we see the solubility of same salt that is potassium iodide at ${{20}^{\circ }}C$ there we have the amount of potassium iodide as $155\,g$ . So, as asked in the question, the extra amount that we need for the two temperatures so we have to find out the difference in amount of potassium iodide at two different temperatures.
Amount of potassium iodide needed $=\,195g\,-155g$
$=40\,g$
Thus we need $40g$ grams of \[KI\]for dissolving in $100g$ of water at two temperature differences.
Note: Saturated solution is formed for a particular temperature. So suppose we want a saturated solution of potassium iodide at ${{40}^{\circ }}C$ it means there is a particular amount of this alt which will dissolve in the water. The moment when no more potassium iodide will dissolve in some amount of water at that particular temperature it will be the saturation point.
Complete step by step answer:
The solubility of different solutes can be seen by using the solubility curve. In the curve we have solubility in $\left( \dfrac{g}{100}c{{m}^{3}}\,water \right)\,Vs\,\left( temperature \right)$ this graph shows how the solubility increases with the increase in temperature. So, see the plot for potassium iodide which is given below.

Here, as we are seeing the solubility of different salts in water and their changes with respect to temperature. So, now see for potassium iodide that at ${{40}^{\circ }}C$ the amount of potassium iodide dissolved in water is $195\,g$ you will able to see through the lines drawn so similarly of we see the solubility of same salt that is potassium iodide at ${{20}^{\circ }}C$ there we have the amount of potassium iodide as $155\,g$ . So, as asked in the question, the extra amount that we need for the two temperatures so we have to find out the difference in amount of potassium iodide at two different temperatures.
Amount of potassium iodide needed $=\,195g\,-155g$
$=40\,g$
Thus we need $40g$ grams of \[KI\]for dissolving in $100g$ of water at two temperature differences.
Note: Saturated solution is formed for a particular temperature. So suppose we want a saturated solution of potassium iodide at ${{40}^{\circ }}C$ it means there is a particular amount of this alt which will dissolve in the water. The moment when no more potassium iodide will dissolve in some amount of water at that particular temperature it will be the saturation point.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




