What is the molecular formula of a diamond?
Answer
562.8k+ views
Hint: Allotropy is the property of any element to exist in more than one form. Diamond, graphite, coke, etc. are called allotropes of carbon. The structure and the position of the atoms in the diamond lattice tell us the molecular formula of diamond and the nature of its bonds.
Complete answer:
Allotropes are the different forms that are possessed by an element that has different structural variations and chemical variations. The most common allotropes can be seen in carbon. Carbon possesses allotropy and has different forms like, graphite, diamond, coke, graphenes, fullerenes, etc. These forms consist of carbon as a sole atom which has different arrangement patterns in their respective crystal lattice.
Diamond is one of the allotropes of carbon that is a crystalline, solid, and homogenous form of carbon. Diamond consists of a 3 – dimensional structure of carbon network. The carbon atoms are $s{{p}^{3}}$ hybridized and form a tetrahedral geometry. Each carbon atom is linked with four other carbon atoms which are again linked with four carbons, which forms a network. These carbons are linked together with covalent bonds. The structure of diamond is:
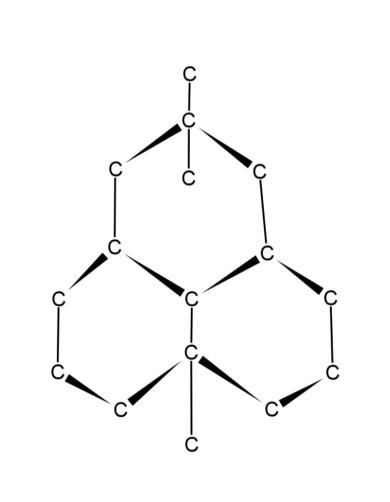
Hence, the molecular formula of diamond is C as it consists of only carbon atoms.
Note:
In diamond carbon is linked with 4 other carbons, so it has a C – 4 arrangement, while in graphite, the carbon atoms are linked with 6 other carbons, so it has a C – 6 arrangement. These are the crystalline allotropes of carbon. While coke, coal and soot are termed as amorphous allotropes of carbon.
Complete answer:
Allotropes are the different forms that are possessed by an element that has different structural variations and chemical variations. The most common allotropes can be seen in carbon. Carbon possesses allotropy and has different forms like, graphite, diamond, coke, graphenes, fullerenes, etc. These forms consist of carbon as a sole atom which has different arrangement patterns in their respective crystal lattice.
Diamond is one of the allotropes of carbon that is a crystalline, solid, and homogenous form of carbon. Diamond consists of a 3 – dimensional structure of carbon network. The carbon atoms are $s{{p}^{3}}$ hybridized and form a tetrahedral geometry. Each carbon atom is linked with four other carbon atoms which are again linked with four carbons, which forms a network. These carbons are linked together with covalent bonds. The structure of diamond is:

Hence, the molecular formula of diamond is C as it consists of only carbon atoms.
Note:
In diamond carbon is linked with 4 other carbons, so it has a C – 4 arrangement, while in graphite, the carbon atoms are linked with 6 other carbons, so it has a C – 6 arrangement. These are the crystalline allotropes of carbon. While coke, coal and soot are termed as amorphous allotropes of carbon.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
Difference Between Prokaryotic Cells and Eukaryotic Cells

Draw a diagram of nephron and explain its structur class 11 biology CBSE

Net gain of ATP in glycolysis a 6 b 2 c 4 d 8 class 11 biology CBSE

Difference between physical and chemical change class 11 chemistry CBSE

Which of the following is the hottest month AMay BJune class 11 social science CBSE

How do I convert ms to kmh Give an example class 11 physics CBSE




