What is the molar mass of ammonium sulfate?
Answer
567.6k+ views
Hint :The molar mass of any chemical compound refers to the ratio of mass of a sample of that particular compound and the amount of substance in that particular sample (in moles). The molar mass is actually a bulk characteristic of a substance rather than molecular. The molar mass is generally represented as $ gmo{l^{ - 1}} $ .
Complete Step By Step Answer:
The molar mass of any compound can be found out by adding the relative atomic masses of each element present in that particular compound. The number of atoms in a compound can be determined from their chemical formula.
Now, let us calculate the molar mass of the given compound i.e. ammonium sulfate. The chemical formula of ammonium sulfate is $ {(N{H_4})_2}S{O_4} $ while its structure is depicted below:
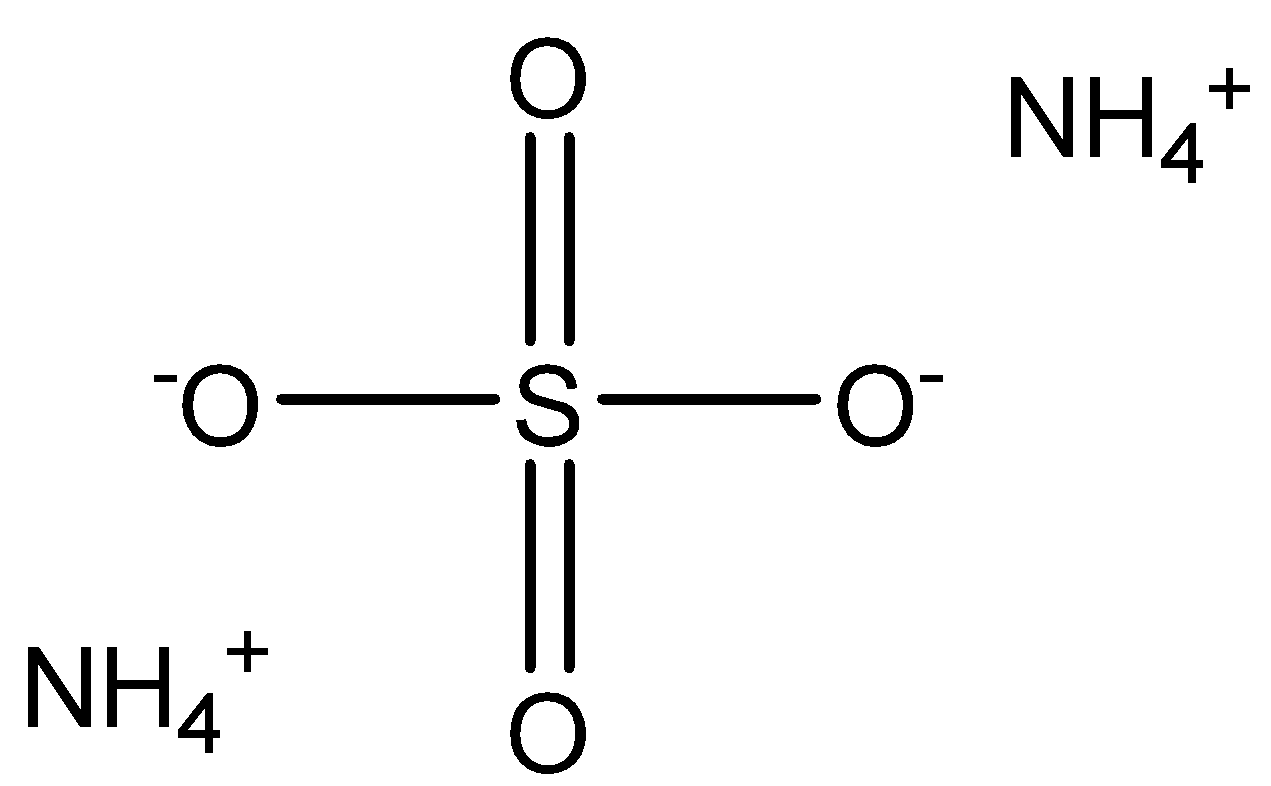
We already know the atomic mass of nitrogen, hydrogen, sulfur and oxygen which is mentioned below:
$ N = 14.0067u \\
H = 1.00784u \\
S = 32.065u \\
O = 15.9994u \\ $
From the chemical formula and structure, it is clear that ammonium sulfate comprises of two atoms of nitrogen, eight atoms of hydrogen, one atom of sulfur and four atoms of oxygen and thus molar mass of this compound can be easily calculated by adding the mass of two nitrogen atoms, eight hydrogen atoms, one sulphur atom and four oxygen atom as shown below:
$ Molar{\text{ }}mass{\text{ }}of{\text{ }}{(N{H_4})_2}S{O_4} = (2 \times N) + (8 \times H) + (1 \times S) + (4 \times O) \\
= (2 \times 14.0067) + (8 \times 1.00784) + (1 \times 32.065) + (4 \times 15.9994) = 132.13872gmo{l^{ - 1}} \\ $
Hence, the molar mass of $ {(N{H_4})_2}S{O_4} $ is 132.13872 $ gmo{l^{ - 1}} $ .
Note :
Molar mass plays a significant role in chemistry especially during setting up an experiment. During testing principles which involve specific amounts or quantities of a substance, molar mass is used to figure out the exact quantity to be weighed of that particular substance. Basically molar mass is used to determine the stoichiometry in the chemical reactions as well as equations.
Complete Step By Step Answer:
The molar mass of any compound can be found out by adding the relative atomic masses of each element present in that particular compound. The number of atoms in a compound can be determined from their chemical formula.
Now, let us calculate the molar mass of the given compound i.e. ammonium sulfate. The chemical formula of ammonium sulfate is $ {(N{H_4})_2}S{O_4} $ while its structure is depicted below:

We already know the atomic mass of nitrogen, hydrogen, sulfur and oxygen which is mentioned below:
$ N = 14.0067u \\
H = 1.00784u \\
S = 32.065u \\
O = 15.9994u \\ $
From the chemical formula and structure, it is clear that ammonium sulfate comprises of two atoms of nitrogen, eight atoms of hydrogen, one atom of sulfur and four atoms of oxygen and thus molar mass of this compound can be easily calculated by adding the mass of two nitrogen atoms, eight hydrogen atoms, one sulphur atom and four oxygen atom as shown below:
$ Molar{\text{ }}mass{\text{ }}of{\text{ }}{(N{H_4})_2}S{O_4} = (2 \times N) + (8 \times H) + (1 \times S) + (4 \times O) \\
= (2 \times 14.0067) + (8 \times 1.00784) + (1 \times 32.065) + (4 \times 15.9994) = 132.13872gmo{l^{ - 1}} \\ $
Hence, the molar mass of $ {(N{H_4})_2}S{O_4} $ is 132.13872 $ gmo{l^{ - 1}} $ .
Note :
Molar mass plays a significant role in chemistry especially during setting up an experiment. During testing principles which involve specific amounts or quantities of a substance, molar mass is used to figure out the exact quantity to be weighed of that particular substance. Basically molar mass is used to determine the stoichiometry in the chemical reactions as well as equations.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




