Minimum $F-S-F$ bond angle is present in:
(a)- $SS{{F}_{2}}$
(b)- $S{{F}_{6}}$
(c)- $S{{F}_{2}}$
(d)- ${{F}_{3}}SSF$
Answer
581.4k+ views
Hint: Bond angle can be measured as the angle between three atoms, keeping one atom as a central atom and it is forming a bond with the other two atoms. The molecule that has many atoms will have a small bond angle because of steric hindrance.
Complete answer:
The bond angle can be measured as the angle between three atoms, keeping one atom as the central atom and it is forming a bond with the other two atoms. So let us examine all the compounds in the options one by one.
(a)- $SS{{F}_{2}}$, this compound is also written as ${{S}_{2}}{{F}_{2}}$ and is known as disulfur difluoride. It has two isomers, so the structure of the isomer that has an $F-S-F$bond is given below:

So there is a lone pair on the sulfur atom so the bond angle is reduced from the tetrahedral angle. So it has $<\text{ 10}{{\text{9}}^{\circ }}\text{28 }\!\!'\!\!\text{ }$ bond angle.
(b)- $S{{F}_{6}}$, this compound is sulfur hexafluoride. This compound has regular octahedral geometry. So its structure is given below:
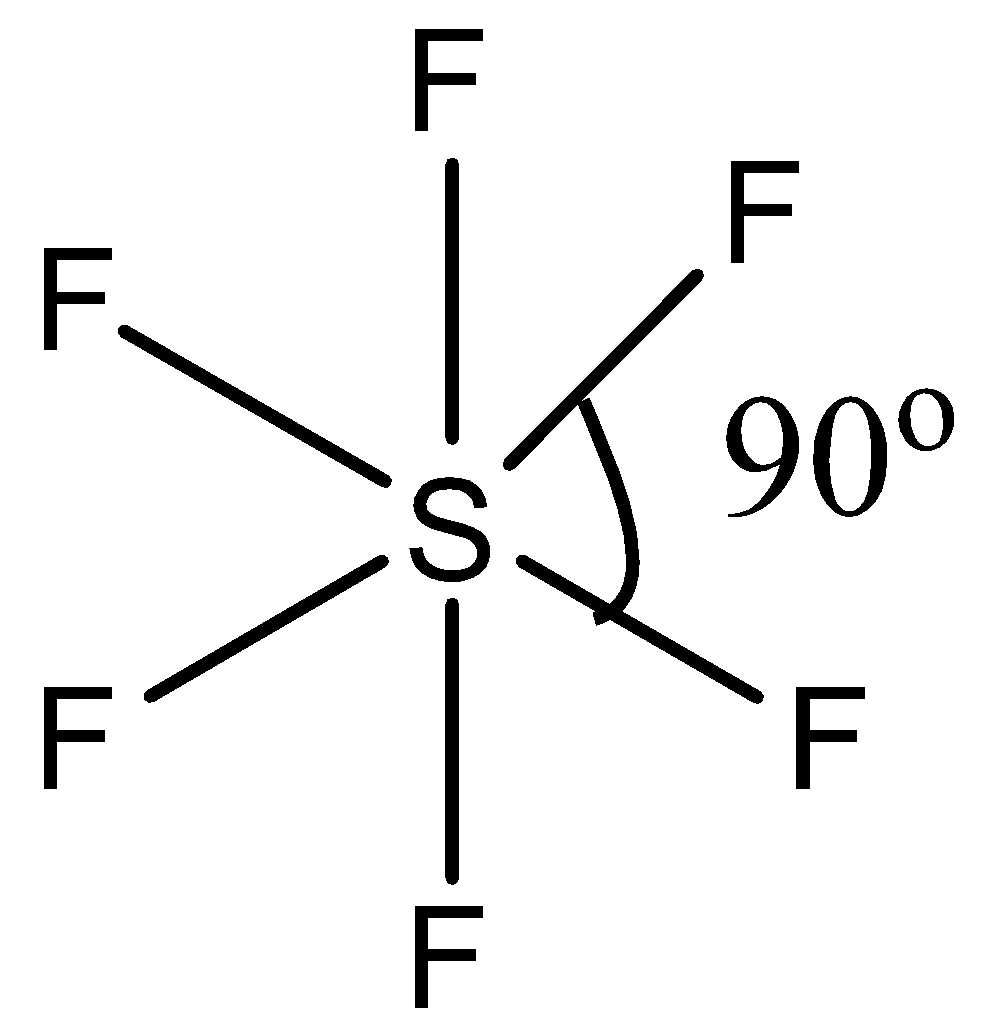
Since it has a symmetric structure, the bond angle is ${{90}^{\circ }}$.
(c)- $S{{F}_{2}}$, this compound is sulfur difluoride. In this compound, the sulfur has two lone pairs. So the structure is given below:

Due to the presence of two lone pairs, the bond angle is ${{98}^{\circ }}$.
(d)- ${{F}_{3}}SSF$, this compound is difluoro disulfur difluoride. Both the sulfur atoms have lone pairs. The structure is given below:

Due to the presence of lone pair the bond is reduced from right angle, i.e., $<\text{ 9}{{\text{0}}^{\circ }}$.
Therefore, the correct answer is an option (d)- ${{F}_{3}}SSF$.
Note:
It must be always noted that if the atom has a lone pair, then there will be repulsion between the bond and lone pairs and the bond angle of the compound will be affected. Lone pair-lone pair repulsion is greater than bond pair-bond pair repulsion.
Complete answer:
The bond angle can be measured as the angle between three atoms, keeping one atom as the central atom and it is forming a bond with the other two atoms. So let us examine all the compounds in the options one by one.
(a)- $SS{{F}_{2}}$, this compound is also written as ${{S}_{2}}{{F}_{2}}$ and is known as disulfur difluoride. It has two isomers, so the structure of the isomer that has an $F-S-F$bond is given below:

So there is a lone pair on the sulfur atom so the bond angle is reduced from the tetrahedral angle. So it has $<\text{ 10}{{\text{9}}^{\circ }}\text{28 }\!\!'\!\!\text{ }$ bond angle.
(b)- $S{{F}_{6}}$, this compound is sulfur hexafluoride. This compound has regular octahedral geometry. So its structure is given below:

Since it has a symmetric structure, the bond angle is ${{90}^{\circ }}$.
(c)- $S{{F}_{2}}$, this compound is sulfur difluoride. In this compound, the sulfur has two lone pairs. So the structure is given below:

Due to the presence of two lone pairs, the bond angle is ${{98}^{\circ }}$.
(d)- ${{F}_{3}}SSF$, this compound is difluoro disulfur difluoride. Both the sulfur atoms have lone pairs. The structure is given below:

Due to the presence of lone pair the bond is reduced from right angle, i.e., $<\text{ 9}{{\text{0}}^{\circ }}$.
Therefore, the correct answer is an option (d)- ${{F}_{3}}SSF$.
Note:
It must be always noted that if the atom has a lone pair, then there will be repulsion between the bond and lone pairs and the bond angle of the compound will be affected. Lone pair-lone pair repulsion is greater than bond pair-bond pair repulsion.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Discuss the various forms of bacteria class 11 biology CBSE




