Lactose on hydrolysis produces a mixture of
(A) Glucose only
(B) Glucose and fructose
(C) Glucose and galactose
(D) Fructose and galactose
Answer
614.7k+ views
Hint: Before finding the product in organic chemistry, always analyse to which category the reactant belongs. Category here refers to the family of the reactant. In organic chemistry, compounds are classified into different families like alcohols, ethers, acids, hydrocarbons, etc. Different families react differently and by different mechanisms.
Complete step by step solution:
-To solve the question, we first need to know what hydrolysis is and what is fructose. Only then can we find the correct product of the reaction. Also we need to find the family of fructose.
-Hydrolysis is a process in which the molecule water is added to a compound and the product obtained is different from that of the reaction. The reaction can be intermolecular or intramolecular.
-The root word for water is hydro and that is why hydrolysis stands for addition of water to the compound. It can be substitution, elimination or addition reaction.
-Sugars are water-soluble carbohydrates. They are sweet in taste and are usually crystalline in nature. They are organic compounds. They are classified as monosaccharides and disaccharides. Monosaccharides are the sugar which are simple and cannot be broken into other compounds. Disaccharides are sugars which can be broken down into simpler monosaccharide sugar molecules.
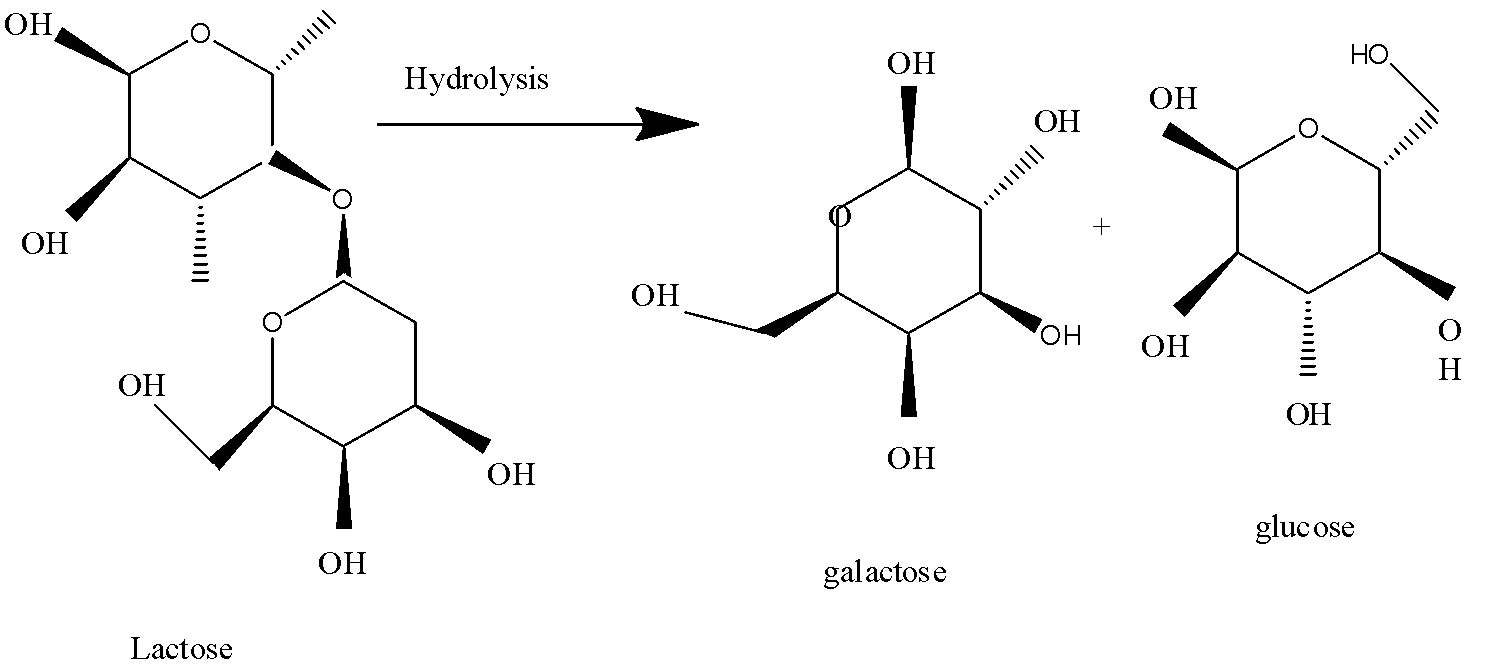
-Now we need to know about lactose first. It is a type of sugar. It is a disaccharide of milk. The process of its hydrolysis is catalysed by an enzyme called lactase just as sucrose hydrolysis is catalysed by sucrase. The chemical formula of lactose is ${{C}_{12}}{{H}_{22}}{{O}_{11}}$ .
-When lactose reacts with water, it forms glucose and galactose. The process is reversible. The formation of lactose can be done easily by the reaction of glucose and galactose molecules, both of which are monosaccharides. The enzyme which increases the rate of reaction is lactase.
Therefore, the correct option is C. glucose and galactose.
Note: The enzyme lactase is present in the human body itself which helps break the complex sugar molecule into simpler sugar molecules to ease the process of digestion. Lactose Is extracted from milk with the help of the enzyme lactase only.
Complete step by step solution:
-To solve the question, we first need to know what hydrolysis is and what is fructose. Only then can we find the correct product of the reaction. Also we need to find the family of fructose.
-Hydrolysis is a process in which the molecule water is added to a compound and the product obtained is different from that of the reaction. The reaction can be intermolecular or intramolecular.
-The root word for water is hydro and that is why hydrolysis stands for addition of water to the compound. It can be substitution, elimination or addition reaction.
-Sugars are water-soluble carbohydrates. They are sweet in taste and are usually crystalline in nature. They are organic compounds. They are classified as monosaccharides and disaccharides. Monosaccharides are the sugar which are simple and cannot be broken into other compounds. Disaccharides are sugars which can be broken down into simpler monosaccharide sugar molecules.
-Now we need to know about lactose first. It is a type of sugar. It is a disaccharide of milk. The process of its hydrolysis is catalysed by an enzyme called lactase just as sucrose hydrolysis is catalysed by sucrase. The chemical formula of lactose is ${{C}_{12}}{{H}_{22}}{{O}_{11}}$ .
-When lactose reacts with water, it forms glucose and galactose. The process is reversible. The formation of lactose can be done easily by the reaction of glucose and galactose molecules, both of which are monosaccharides. The enzyme which increases the rate of reaction is lactase.
Therefore, the correct option is C. glucose and galactose.

Note: The enzyme lactase is present in the human body itself which helps break the complex sugar molecule into simpler sugar molecules to ease the process of digestion. Lactose Is extracted from milk with the help of the enzyme lactase only.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




