
In the cyclo-${{S}_{8}}$ molecule of rhombic sulphur, all the S-S bond lengths and all the S-S-S bond angles are respectively (give approximate values)
(A) 204 pm and ${{105}^{{}^\circ }}$
(B) 102 pm ${{120}^{{}^\circ }}$
(C) 204 pm ${{180}^{{}^\circ }}$
(D) 102 pm ${{60}^{{}^\circ }}$
Answer
592.2k+ views
Hint: Sulphur forms numerous allotropes, but let us study the two most important allotropes of sulphur-yellow rhombic sulphur ($\alpha $-sulphur) and the monoclinic ($\beta $-sulphur). The allotropes of sulphur are inter-convertible i.e. rhombic sulphur when heated above 369K gives monoclinic sulphur.
Complete step by step answer:
Octasulfur is an inorganic chemical with the chemical formula ${{S}_{8}}$. It is a yellow solid, and is odourless and tasteless. It is the most common allotrope of sulfur. It is a major industrial chemical that occurs widely in nature.
${{S}_{8}}$ has a puckered ring structure where each sulphur atom is bonded with two other atoms forming a ring. In ${{S}_{8}}$the bond angle between two bonds of one sulphur atom is relatively small and they can withstand because of the larger size of sulphur atom which makes repulsion between them weaker.
${{S}_{8}}$ molecule has eight single covalent bonds among eight S atoms and form a closed crown shaped structure.The structure of ${{S}_{8}}$ molecule is as follows:
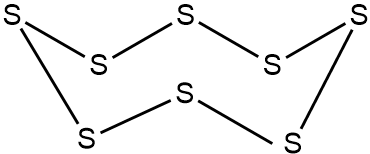
In all of the ring forms of S, the S-S distance is 204 pm and the bond angle of S-S-S is ${{105}^{{}^\circ }}$.
So, the correct answer is “Option A”.
Note: They are all soluble in $C{{S}_{2}}$. The Orthorhombic form of sulphur is thermodynamically and most stable. Rhombic sulphur is prepared by dissolving powdered sulphur in carbon disulphide at room temperature. The mixture is then filtered. The filtrate is then kept in a small beaker covered with a filter paper.
Complete step by step answer:
Octasulfur is an inorganic chemical with the chemical formula ${{S}_{8}}$. It is a yellow solid, and is odourless and tasteless. It is the most common allotrope of sulfur. It is a major industrial chemical that occurs widely in nature.
${{S}_{8}}$ has a puckered ring structure where each sulphur atom is bonded with two other atoms forming a ring. In ${{S}_{8}}$the bond angle between two bonds of one sulphur atom is relatively small and they can withstand because of the larger size of sulphur atom which makes repulsion between them weaker.
${{S}_{8}}$ molecule has eight single covalent bonds among eight S atoms and form a closed crown shaped structure.The structure of ${{S}_{8}}$ molecule is as follows:

In all of the ring forms of S, the S-S distance is 204 pm and the bond angle of S-S-S is ${{105}^{{}^\circ }}$.
So, the correct answer is “Option A”.
Note: They are all soluble in $C{{S}_{2}}$. The Orthorhombic form of sulphur is thermodynamically and most stable. Rhombic sulphur is prepared by dissolving powdered sulphur in carbon disulphide at room temperature. The mixture is then filtered. The filtrate is then kept in a small beaker covered with a filter paper.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which country won the ICC Men's ODI World Cup in 2023?

In cricket, how many legal balls are there in a standard over?

Explain the Treaty of Vienna of 1815 class 10 social science CBSE

A boat goes 24 km upstream and 28 km downstream in class 10 maths CBSE

What does "powerplay" mean in limited-overs cricket?

What is the "Powerplay" in T20 cricket?




