In pyrrole the electron density is maximum on which number of atoms?
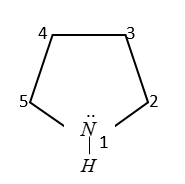
(A) 2 and 3
(B) 3 and 4
(C) 2 and 4
(D) 2 and 5

Answer
608.1k+ views
Hint: In conjugation system $\pi $electrons and lone pair of electrons are in conjugation with each other. $\pi $electrons are delocalized and constantly moving on aromatic rings.
Step by step answer: Pyrol is an aromatic compound. Let us prove that pyrrole is an aromatic compound.
These compounds should have the following properties.
(i) Each carbon atom should be planer ($s{p^2}$ hybridization)
Here C-atoms at 2, 3, 4, 5 positions are $s{p^2}$ hybridized
(ii) There should be a conjugated system.
Conjugated system has $\pi $-bond -$\sigma $-bond-lone pair of electrons alternately.
This system also presents in pyrol.
(iii) There should be $(4n + 2)\pi $electrons present
Where $n = 0,1,2,3 - - - - - $
i.e., $2\pi $ electrons, $6\pi $ electrons, $10\pi $ electrons and so on.
$\therefore $ Pyrol is an aromatic compound.
In aromatic compounds $\pi $ electrons are delocalized.
This delocalization of $\pi $ electrons is with a lone pair of electrons.
$\therefore $ In pyrol, electron density is maximum at 2nd and $5m$ carbon.
We can observe this from its Resonance structure. These structures are stable, other structures are not stable.
Therefore, from the above explanation the correct option is (D) 2 and 5.
Note: Electron density of aromatic compound can be determined by deallocation of $\pi $electrons and lone pair of electrons on aromatic rings.
Step by step answer: Pyrol is an aromatic compound. Let us prove that pyrrole is an aromatic compound.
These compounds should have the following properties.
(i) Each carbon atom should be planer ($s{p^2}$ hybridization)
Here C-atoms at 2, 3, 4, 5 positions are $s{p^2}$ hybridized
(ii) There should be a conjugated system.
Conjugated system has $\pi $-bond -$\sigma $-bond-lone pair of electrons alternately.
This system also presents in pyrol.
(iii) There should be $(4n + 2)\pi $electrons present
Where $n = 0,1,2,3 - - - - - $
i.e., $2\pi $ electrons, $6\pi $ electrons, $10\pi $ electrons and so on.
$\therefore $ Pyrol is an aromatic compound.
In aromatic compounds $\pi $ electrons are delocalized.
This delocalization of $\pi $ electrons is with a lone pair of electrons.

$\therefore $ In pyrol, electron density is maximum at 2nd and $5m$ carbon.
We can observe this from its Resonance structure. These structures are stable, other structures are not stable.
Therefore, from the above explanation the correct option is (D) 2 and 5.
Note: Electron density of aromatic compound can be determined by deallocation of $\pi $electrons and lone pair of electrons on aromatic rings.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




