In bisulphate ion, the formal charge on the sulfur atom is:
A. \[+1\]
B. \[+2\]
C. \[+4\]
D. \[+6\]
Answer
592.8k+ views
Hint: Formal charge of an atom in a molecule can be found by the relation: $formal\,charge=valence\text{ }electrons-\left( non-bonding\text{ }valance\text{ }electrons \right)-\dfrac{\left( bonding\text{ }electrons \right)}{2}$
Using the above relation, find the formal charge on the sulfur atom in bisulphate ion.
Complete step by step answer:
We know that the relation to find the formal charge mathematical can be expressed as shown below:
$formal\,charge=valence\text{ }electrons-\left( non-bonding\text{ }valance\text{ }electrons \right)-\dfrac{\left( bonding\text{ }electrons \right)}{2}$
The structure of the bisulphate ion is as follows:
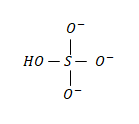
In which the middle atom is the sulfur atom.
The valence electrons of the sulfur atom is = 06 electrons;
The non-bonding valence electrons of the sulfur atom in the above molecule is = 0 electrons;
The bonding valence electrons of the sulfur atom in the above molecule is = 08 electrons.
Put all these values in the above formula and calculate the formal charge
After substituting all the values in the above relation we obtain
$formal\,charge=6-\left( 0 \right)-\dfrac{8}{2}\text{ = +2 }$
Therefore the required formal charge on the sulfur atom in the bisulphate molecule is \[+2\]
Hence option (B) is the correct answer.
Note: Formal charge can be defined as the charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms, regardless of the relative electronegativity.
The formal charge on an atom in a molecule reflects the electron count associated with the atom compared to the isolated neutral atom.
Using the above relation, find the formal charge on the sulfur atom in bisulphate ion.
Complete step by step answer:
We know that the relation to find the formal charge mathematical can be expressed as shown below:
$formal\,charge=valence\text{ }electrons-\left( non-bonding\text{ }valance\text{ }electrons \right)-\dfrac{\left( bonding\text{ }electrons \right)}{2}$
The structure of the bisulphate ion is as follows:
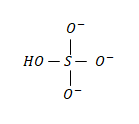
In which the middle atom is the sulfur atom.
The valence electrons of the sulfur atom is = 06 electrons;
The non-bonding valence electrons of the sulfur atom in the above molecule is = 0 electrons;
The bonding valence electrons of the sulfur atom in the above molecule is = 08 electrons.
Put all these values in the above formula and calculate the formal charge
After substituting all the values in the above relation we obtain
$formal\,charge=6-\left( 0 \right)-\dfrac{8}{2}\text{ = +2 }$
Therefore the required formal charge on the sulfur atom in the bisulphate molecule is \[+2\]
Hence option (B) is the correct answer.
Note: Formal charge can be defined as the charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms, regardless of the relative electronegativity.
The formal charge on an atom in a molecule reflects the electron count associated with the atom compared to the isolated neutral atom.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Class 11 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Accountancy: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

In what year Guru Nanak Dev ji was born A15 April 1469 class 11 social science CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE




