Given, \[\text{PhC}{{\text{H}}_{\text{2}}}\text{C}{{\text{H}}_{\text{3}}}\xrightarrow[\text{(ii)}{{\text{H}}_{\text{3}}}{{\text{O}}^{\text{+}}}]{\text{(i)Cr}{{\text{O}}_{\text{2}}}\text{C}{{\text{1}}_{\text{2}}}\text{/CC}{{\text{l}}_{\text{4}}}}\text{PhC}{{\text{H}}_{\text{2}}}\text{CHO}\]
The above reaction is an example of which name reaction?
A) Rosenmund reduction
B) Birch reduction
C) Mendius reduction
D) Étard reduction
Answer
615k+ views
Hint: The chromyl chloride is an oxidizing agent. It oxidizes the benzylic methyl group by the removal of hydrogen from the alkyl group. The oxygen of chromyl chloride forms a complex called the Étard complex. This complex undergoes the sigmatropic rearrangement to form an aldehyde.
Complete step by step answer:
The reaction which offers a way to oxidize the aromatic methyl’s group $\text{(Ar-C}{{\text{H}}_{\text{3}}}\text{)}$ into an aldehyde $\text{(Ar-CHO)}$ using a chromyl chloride\[\text{Cr}{{\text{O}}_{\text{2}}}\text{C}{{\text{l}}_{\text{2}}}\]. This oxidation is called the Étard oxidation.
The reaction starts with a reaction of alkene which is formed by the removal of hydrogen and the allylic hydrogen which further reacts with the chromyl chloride to give an Étard complex. This complex is then decomposed by reducing the environment to avoid further oxidation of the aldehyde to a carboxylic acid. Carbon tetrachloride is the most commonly used solvent. Aldehyde obtained by the Étard oxidation is high purity.
The chromyl chloride forms an Étard complex.
An end reaction with chromyl chloride forming a precipitated Étard Complex is the first step of this mechanism.
The sigmatropic reaction is a pericyclic reaction where one$\text{ }\!\!\sigma\!\!\text{ }$ bond is converted into the other $\text{ }\!\!\sigma\!\!\text{ }$ bond. The methyl is converted into the aldehyde through the $\left[ 2,3 \right]$ sigmatropic rearrangement reaction.
The Étard complex formed is decomposed into the Phenyl acetaldehyde.
This is a very easy method for the oxidation of ethylbenzene into the Phenyl acetaldehyde.
Thus the given reaction,
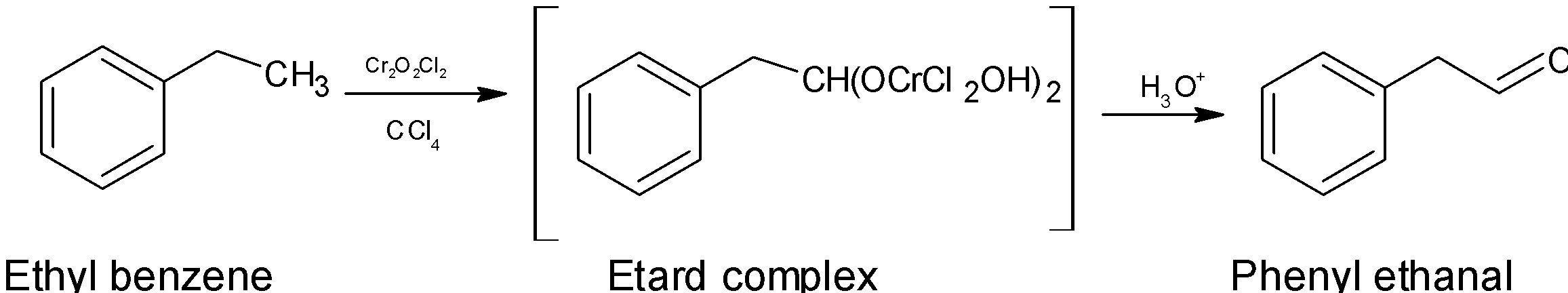
The conversion of ethylbenzene to Phenyl acetaldehyde is Étard oxidation.
So, the correct answer is “Option D”.
Note: The Étard complex can undergo the rapid oxidation to give carboxylic acid, therefore used as a reducing agent to decompose complex.
Sigmatropic rearrangement makes it difficult for the other alkyl benzene to formaldehyde.
Complete step by step answer:
The reaction which offers a way to oxidize the aromatic methyl’s group $\text{(Ar-C}{{\text{H}}_{\text{3}}}\text{)}$ into an aldehyde $\text{(Ar-CHO)}$ using a chromyl chloride\[\text{Cr}{{\text{O}}_{\text{2}}}\text{C}{{\text{l}}_{\text{2}}}\]. This oxidation is called the Étard oxidation.
The reaction starts with a reaction of alkene which is formed by the removal of hydrogen and the allylic hydrogen which further reacts with the chromyl chloride to give an Étard complex. This complex is then decomposed by reducing the environment to avoid further oxidation of the aldehyde to a carboxylic acid. Carbon tetrachloride is the most commonly used solvent. Aldehyde obtained by the Étard oxidation is high purity.
The chromyl chloride forms an Étard complex.
An end reaction with chromyl chloride forming a precipitated Étard Complex is the first step of this mechanism.
The sigmatropic reaction is a pericyclic reaction where one$\text{ }\!\!\sigma\!\!\text{ }$ bond is converted into the other $\text{ }\!\!\sigma\!\!\text{ }$ bond. The methyl is converted into the aldehyde through the $\left[ 2,3 \right]$ sigmatropic rearrangement reaction.
The Étard complex formed is decomposed into the Phenyl acetaldehyde.
This is a very easy method for the oxidation of ethylbenzene into the Phenyl acetaldehyde.
Thus the given reaction,

The conversion of ethylbenzene to Phenyl acetaldehyde is Étard oxidation.
So, the correct answer is “Option D”.
Note: The Étard complex can undergo the rapid oxidation to give carboxylic acid, therefore used as a reducing agent to decompose complex.
Sigmatropic rearrangement makes it difficult for the other alkyl benzene to formaldehyde.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




