For \[{{H}_{3}}P{{O}_{3}}\]and \[{{H}_{3}}P{{O}_{4}}\] the correct choice is:
A. \[{{H}_{3}}P{{O}_{3}}\] is dibasic and reducing.
B. \[{{H}_{3}}P{{O}_{3}}\] is dibasic and non-reducing.
C. \[{{H}_{3}}P{{O}_{4}}\] is tribasic and reducing.
D. \[{{H}_{3}}P{{O}_{4}}\] is tribasic and non-reducing.
Answer
615k+ views
Hint: Phosphoric acids are strong acids. It has 3 hydrogen atoms but only 2 are isonisable. Thus it is soluble in water.
Complete step by step solution:
Phosphorus acid or phosphonic acid is a colourless deliquescent crystalline solid which is highly acidic in nature. Even though it has 3 hydrogen, only 2 are ionisable. Hence it is dibasic acid and ionises as
\[{{H}_{3}}P{{O}_{3}}\to {{H}^{+}}+{{H}_{2}}PO_{3}^{-}\]
\[{{H}_{3}}PO_{3}^{-}\to {{H}^{+}}+{{H}_{2}}PO_{3}^{2-}\]
The other hydrogen, which does not take part in ionisation, has a reducing nature. Thus \[{{H}_{3}}P{{O}_{3}}\] act as a reducing agent. So the correct answer for the question is (a).
-Phosphorus acid is obtained by hydrolysis of phosphorus trichloride and phosphorus trioxide.
-Phosphorus acid and its salts are strong reducing agents, as they are readily oxidisable to phosphoric acid and phosphates, respectively.
\[HPO_{3}^{2-}+3O{{H}^{-}}\to PO_{4}^{3-}+2{{H}_{2}}O+2{{e}^{-}}\]
- Phosphorus acid has the capability of reducing salts of copper, silver, gold etc., to their respective metals.
-The structure of phosphorus acid is shown as
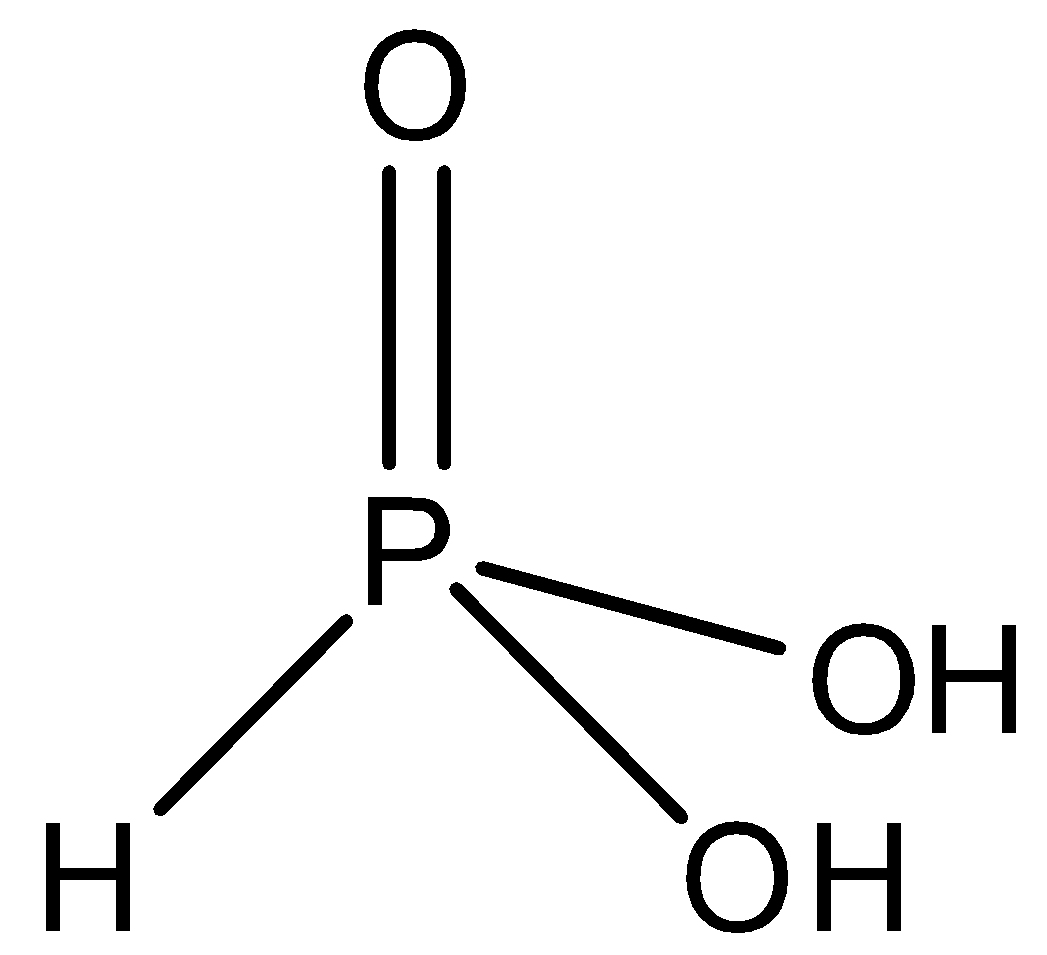
-Orthophosphoric acid \[{{H}_{3}}P{{O}_{4}}\] is a tribasic weak acid. It also has 3 hydrogen atoms, but has 4 oxygen atoms. It is not a reducing agent because all the 3 of its hydrogen ionises in water. It structure is shown as
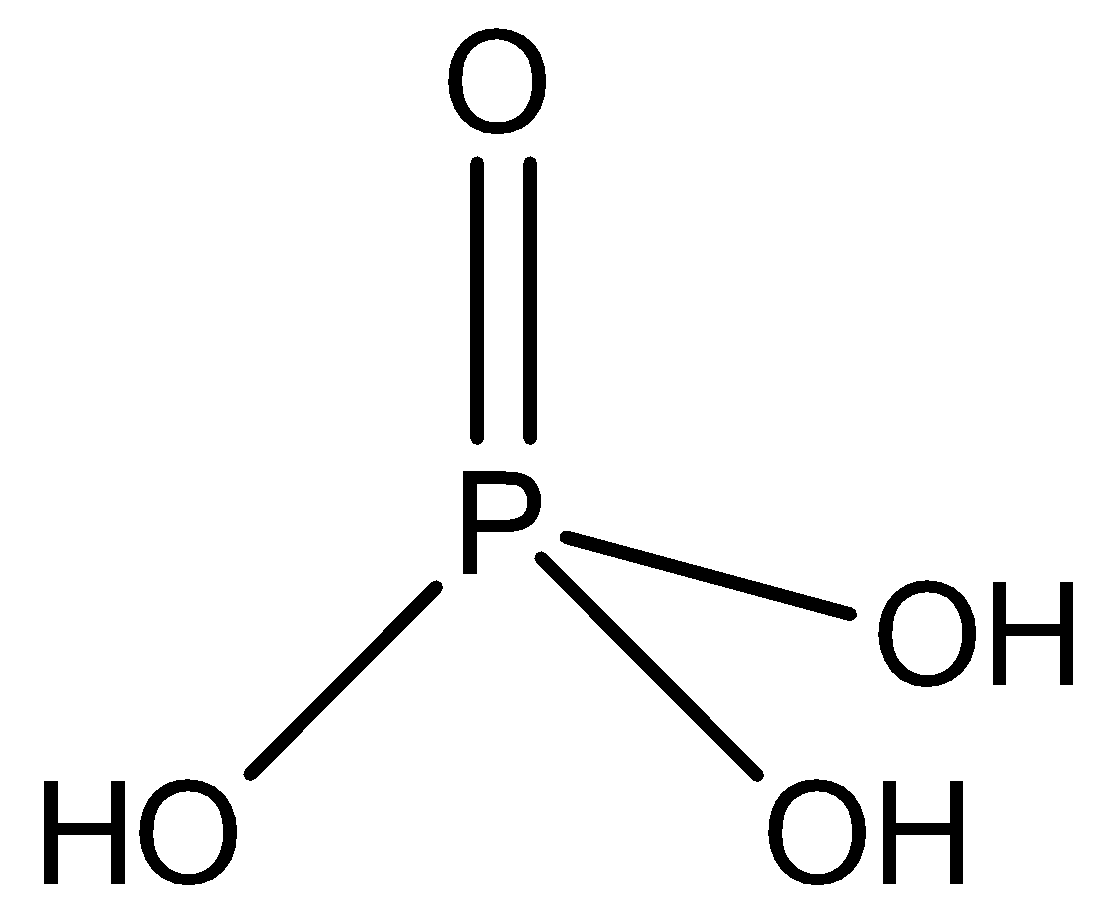
-Phosphoric acid is a syrup liquid which combines with water gives a semi hydrate \[2{{H}_{3}}P{{O}_{4}}.{{H}_{2}}O\]
-It ionises in 3 stages
\[{{H}_{3}}PO_{4}^{{}}\rightleftharpoons {{H}^{+}}+{{H}_{2}}PO_{4}^{-}\]
\[{{H}_{3}}PO_{4}^{-}\rightleftharpoons {{H}^{+}}+{{H}_{2}}PO_{4}^{2-}\]
\[{{H}_{3}}PO_{4}^{2-}\rightleftharpoons {{H}^{+}}+PO_{4}^{3-}\]
Its first ionisation takes place quite fast, but the second and third ionisation takes place very slow. It forms a series of salts.
Thus, the correct answer is option (a), Phosphorus acid is dibasic and reducing.
Note: There are chances that we assume even phosphoric acid is reducing like phosphorus acid. But it is not a reducing agent.
Complete step by step solution:
Phosphorus acid or phosphonic acid is a colourless deliquescent crystalline solid which is highly acidic in nature. Even though it has 3 hydrogen, only 2 are ionisable. Hence it is dibasic acid and ionises as
\[{{H}_{3}}P{{O}_{3}}\to {{H}^{+}}+{{H}_{2}}PO_{3}^{-}\]
\[{{H}_{3}}PO_{3}^{-}\to {{H}^{+}}+{{H}_{2}}PO_{3}^{2-}\]
The other hydrogen, which does not take part in ionisation, has a reducing nature. Thus \[{{H}_{3}}P{{O}_{3}}\] act as a reducing agent. So the correct answer for the question is (a).
-Phosphorus acid is obtained by hydrolysis of phosphorus trichloride and phosphorus trioxide.
-Phosphorus acid and its salts are strong reducing agents, as they are readily oxidisable to phosphoric acid and phosphates, respectively.
\[HPO_{3}^{2-}+3O{{H}^{-}}\to PO_{4}^{3-}+2{{H}_{2}}O+2{{e}^{-}}\]
- Phosphorus acid has the capability of reducing salts of copper, silver, gold etc., to their respective metals.
-The structure of phosphorus acid is shown as

-Orthophosphoric acid \[{{H}_{3}}P{{O}_{4}}\] is a tribasic weak acid. It also has 3 hydrogen atoms, but has 4 oxygen atoms. It is not a reducing agent because all the 3 of its hydrogen ionises in water. It structure is shown as

-Phosphoric acid is a syrup liquid which combines with water gives a semi hydrate \[2{{H}_{3}}P{{O}_{4}}.{{H}_{2}}O\]
-It ionises in 3 stages
\[{{H}_{3}}PO_{4}^{{}}\rightleftharpoons {{H}^{+}}+{{H}_{2}}PO_{4}^{-}\]
\[{{H}_{3}}PO_{4}^{-}\rightleftharpoons {{H}^{+}}+{{H}_{2}}PO_{4}^{2-}\]
\[{{H}_{3}}PO_{4}^{2-}\rightleftharpoons {{H}^{+}}+PO_{4}^{3-}\]
Its first ionisation takes place quite fast, but the second and third ionisation takes place very slow. It forms a series of salts.
Thus, the correct answer is option (a), Phosphorus acid is dibasic and reducing.
Note: There are chances that we assume even phosphoric acid is reducing like phosphorus acid. But it is not a reducing agent.
Recently Updated Pages
Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Explain the structure of megasporangium class 12 biology CBSE

What are the major means of transport Explain each class 12 social science CBSE

How many chromosomes are found in human ovum a 46 b class 12 biology CBSE

The diagram of the section of a maize grain is given class 12 biology CBSE




