Draw the branched structural formula of:
(A) Methoxy methane.
(B) $ 2- $ methylpropan $ -2- $ ol.
Answer
552k+ views
Hint :We know that for this problem, we should know about the general points of the IUPAC naming while naming the compound like the naming of functional groups, alkyl groups, chains, etc. So that we can easily draw the structure of the molecule by using its name.
Complete Step By Step Answer:
In the given question we have to draw the structural formula of the given compound that is a $ 2- $ methylpropan $ -2- $ ol and Methoxy methane molecule. IUPAC or International Union of Pure and Applied Compounds are a method of naming any organic compound. There are some points to be remembered while names such as: If there is one carbon atom in the longest chain then it is named as meth, if two carbon atoms are present then it is named as eth and if three carbon atoms are present then it is named as a prop. So, as we can see that the main chain is named as 'prop' so it consists of three carbon atoms with one methyl group that is one carbon atom at $ 2- $ carbon.
Now, the word 'ane' means that there is a single bond present only because for double bond 'ene' is used and for a triple bond, we use 'yne'. Now, the last one is the functional group 'ol' which is a suffix for an alcohol group. So, both the structural formula of the given compound will be:
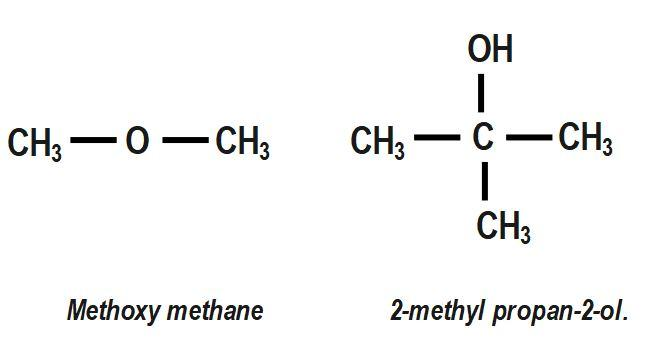
Note :
Remember that for other functional groups such as carboxylic acid the suffix -oic acid is used, for aldehyde -al is used, etc. The naming of the functional group is also dependent on the priority order of the functional groups.
Complete Step By Step Answer:
In the given question we have to draw the structural formula of the given compound that is a $ 2- $ methylpropan $ -2- $ ol and Methoxy methane molecule. IUPAC or International Union of Pure and Applied Compounds are a method of naming any organic compound. There are some points to be remembered while names such as: If there is one carbon atom in the longest chain then it is named as meth, if two carbon atoms are present then it is named as eth and if three carbon atoms are present then it is named as a prop. So, as we can see that the main chain is named as 'prop' so it consists of three carbon atoms with one methyl group that is one carbon atom at $ 2- $ carbon.
Now, the word 'ane' means that there is a single bond present only because for double bond 'ene' is used and for a triple bond, we use 'yne'. Now, the last one is the functional group 'ol' which is a suffix for an alcohol group. So, both the structural formula of the given compound will be:

Note :
Remember that for other functional groups such as carboxylic acid the suffix -oic acid is used, for aldehyde -al is used, etc. The naming of the functional group is also dependent on the priority order of the functional groups.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




