Draw a dot structure of \[B{r_3}^{ - 1}\]. Deduce approximate value of bond angle.
Answer
551.1k+ views
Hint: Bromine is from group7 or 17, so it has valence electron seven. We have been given three bromines in question and then there is an additional negative sign indicating additional charge on the structure. So, in total we have three bromines with charge on each is 7.
Complete answer:
The Lewis dot structure is-
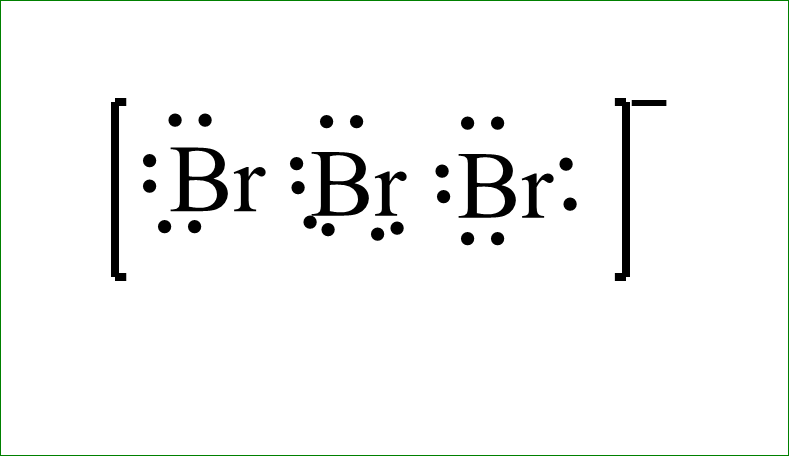
The bond angle is \[{180^ \circ }\], since it is linear in shape. Which makes it twenty-one and additional one negative charge makes it twenty-two electrons. We will line them up in such a way that they get symmetry. We will take the electrons we have and we will place the two electrons between atoms to form a chemical bond. Then we will draw the electrons outside of atoms to fill their octets. We have about 2,4,6,8,16 and then back to the central bromine.,18 and 20. So everything has octets now, but we only use 20. We have additional two valence electrons left which we will place somewhere.
Since bromine is below period second, in the periodic table, that means, it can have an expanded octet. It can have more than eight electrons. So, we will put these last two valence electrons on the central bromine. Now we have used all the twenty-two valence electrons.
It is lowest for fluorine. Hence, the most powerful oxidising agent is fluorine.
Note:
The inner bromine has ten valence electrons but that is okay, because bromine can have expanded octet. The lewis dot structure will be put inside the brackets and the negative sign will be indicated outside it.
Complete answer:
The Lewis dot structure is-
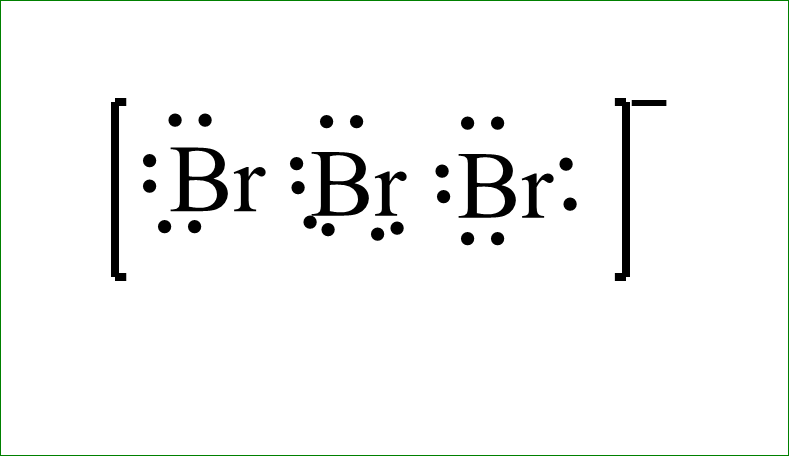
The bond angle is \[{180^ \circ }\], since it is linear in shape. Which makes it twenty-one and additional one negative charge makes it twenty-two electrons. We will line them up in such a way that they get symmetry. We will take the electrons we have and we will place the two electrons between atoms to form a chemical bond. Then we will draw the electrons outside of atoms to fill their octets. We have about 2,4,6,8,16 and then back to the central bromine.,18 and 20. So everything has octets now, but we only use 20. We have additional two valence electrons left which we will place somewhere.
Since bromine is below period second, in the periodic table, that means, it can have an expanded octet. It can have more than eight electrons. So, we will put these last two valence electrons on the central bromine. Now we have used all the twenty-two valence electrons.
It is lowest for fluorine. Hence, the most powerful oxidising agent is fluorine.
Note:
The inner bromine has ten valence electrons but that is okay, because bromine can have expanded octet. The lewis dot structure will be put inside the brackets and the negative sign will be indicated outside it.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE




