Cyclopentadienyl anion is:
A. Benzenoid and aromatic
B. Non-benzenoid and aromatic
C. Non-benzenoid and non-aromatic
D. Non-benzenoid and anti-aromatic
Answer
558.6k+ views
Hint: Cyclopentadienyl is present in a form of free radical in the shape of five membered ring and have the molecular formula $(4n+2)\pi $${{C}_{5}}{{H}_{5}}$and cyclopentadienyl anion is shown by the molecular formula ${{C}_{5}}{{H}_{5}}^{-}$and abbreviated as $C{{p}^{-}}$.
Complete answer:
To be an aromatic compound, a compound must follow some conditions:
1. It should be cyclic in nature and the compound given contains a 5-membered cyclic ring.
2. Every atom in the ring must be conjugated which delocalize the pi electron system.
3. The compound must follow Huckel’s rule i.e. $(4n+2)\pi $electrons i.e. the total number of electrons present should follows this rule and n can be any integer i.e. 1,2, 3….. but always have a whole number.
In the given compound anion contributes two more electrons to the compound and 4 pi electrons are already so we can say that it follows Huckel’s rule with 6$\pi $electrons where n is 1.
4. Molecules must be planar as we know that planar molecules possess higher potential energy.
Given compound follows all the properties of being aromatic, so it is aromatic in nature on the other hand the compound is said to be benzenoid if it contains benzene ring but it contains a five-membered ring so non-benzenoid in nature. This can be shown as follows:
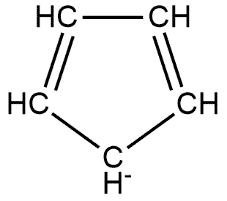
Hence option B is the correct answer.
Note:
Cyclopentadienyl anion is formed by the deprotonation of the molecule cyclopentadienyl. On the other hand if we take the case of cyclopentadienyl cation then it does not follow Huckel’s rule as there are only 4$\pi $electrons present so it becomes anti-aromatic in nature.
Complete answer:
To be an aromatic compound, a compound must follow some conditions:
1. It should be cyclic in nature and the compound given contains a 5-membered cyclic ring.
2. Every atom in the ring must be conjugated which delocalize the pi electron system.
3. The compound must follow Huckel’s rule i.e. $(4n+2)\pi $electrons i.e. the total number of electrons present should follows this rule and n can be any integer i.e. 1,2, 3….. but always have a whole number.
In the given compound anion contributes two more electrons to the compound and 4 pi electrons are already so we can say that it follows Huckel’s rule with 6$\pi $electrons where n is 1.
4. Molecules must be planar as we know that planar molecules possess higher potential energy.
Given compound follows all the properties of being aromatic, so it is aromatic in nature on the other hand the compound is said to be benzenoid if it contains benzene ring but it contains a five-membered ring so non-benzenoid in nature. This can be shown as follows:

Hence option B is the correct answer.
Note:
Cyclopentadienyl anion is formed by the deprotonation of the molecule cyclopentadienyl. On the other hand if we take the case of cyclopentadienyl cation then it does not follow Huckel’s rule as there are only 4$\pi $electrons present so it becomes anti-aromatic in nature.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




