Chlorobenzene can be converted to benzene by treatment with $NaOH/CaO$.
(a) True
(b) False
Answer
596.4k+ views
Hint: When chlorobenzene is made to react with the sodium hydroxide or the calcium oxide, it results in the formation of the compound which is aromatic in nature and consists of oxygen and sodium atoms attached to it. Now with the help of this, you can easily identify whether the given statement is correct or wrong.
Complete answer:
First of all, let’s discuss chlorobenzene. Chlorobenzene is an aromatic compound. When chlorine is attached to the benzene ring, then the aromatic compound is known as chlorobenzene.
Now considering the statement;
When chlorobenzene is made to undergo reaction with the sodium hydroxide i.e. NaOH , it results in the formation of the sodium phenoxide. The reaction is supposed to occur as;
${{C}_{6}}{{H}_{5}}Cl+NaOH\to {{C}_{6}}{{H}_{5}}ONa$
So, thus Chlorobenzene cannot be converted to benzene by treatment with $NaOH/CaO$ and the given statement is false.
Hence, option (b) is correct.
Additional information:
Chlorobenzene, itself, is formed by the halogenation of the benzene ring in the presence of light. The benzene ring undergoes electrophilic substitution reaction and the hydrogen atom of the benzene ring is replaced by the chlorine atom, thus, resulting in the formation of the chlorobenzene. The reaction is supposed to occur as-
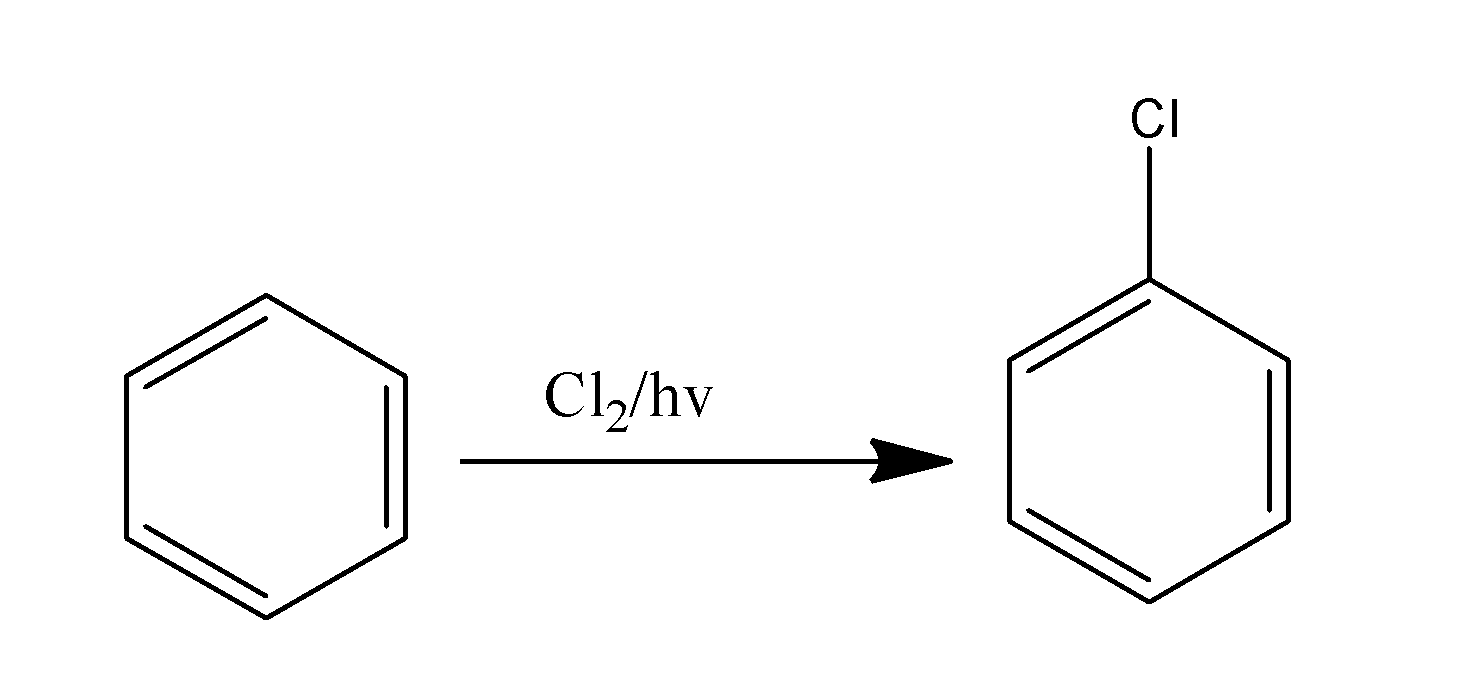
Note:
Chlorobenzene when is treated with the hydrogen in the presence of nickel alloy and sodium hydroxide, it results in the formation of the benzene ring. The reaction is supposed to occur as;
$\,{{C}_{6}}{{H}_{5}}Cl+2H\xrightarrow[NaOH]{Ni-alloy}{{C}_{6}}{{H}_{6}}+HCl$
Complete answer:
First of all, let’s discuss chlorobenzene. Chlorobenzene is an aromatic compound. When chlorine is attached to the benzene ring, then the aromatic compound is known as chlorobenzene.
Now considering the statement;
When chlorobenzene is made to undergo reaction with the sodium hydroxide i.e. NaOH , it results in the formation of the sodium phenoxide. The reaction is supposed to occur as;
${{C}_{6}}{{H}_{5}}Cl+NaOH\to {{C}_{6}}{{H}_{5}}ONa$
So, thus Chlorobenzene cannot be converted to benzene by treatment with $NaOH/CaO$ and the given statement is false.
Hence, option (b) is correct.
Additional information:
Chlorobenzene, itself, is formed by the halogenation of the benzene ring in the presence of light. The benzene ring undergoes electrophilic substitution reaction and the hydrogen atom of the benzene ring is replaced by the chlorine atom, thus, resulting in the formation of the chlorobenzene. The reaction is supposed to occur as-

Note:
Chlorobenzene when is treated with the hydrogen in the presence of nickel alloy and sodium hydroxide, it results in the formation of the benzene ring. The reaction is supposed to occur as;
$\,{{C}_{6}}{{H}_{5}}Cl+2H\xrightarrow[NaOH]{Ni-alloy}{{C}_{6}}{{H}_{6}}+HCl$
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




