What is the chemical formula of sodium carbonate?
A. \[Na(C{{O}_{3}})\]
B. \[Na{{(C{{O}_{3}})}_{2}}\]
C. \[N{{a}_{2}}(C{{O}_{3}})\]
D. \[N{{a}_{2}}{{(C{{O}_{3}})}_{3}}\]
Answer
639.9k+ views
Hint: In sodium carbonate (other names are washing soda, soda ash or soda crystals), the charge on the sodium cation is +1, the charge on the carbonate anion is -2. So, balance and construct the chemical formula of sodium carbonate with the help of the respective valencies of the ions.
Complete step by step answer:
The valency of a complex ion is the same as the value of the charge of the ion.
Carbonate (\[C{{O}_{3}}^{2-}\]) has a charge of -2, so valency is 2. Valency of sodium (\[N{{a}^{+}}\]) is 1.
Hence, as per crossover rule, crossing valency over:
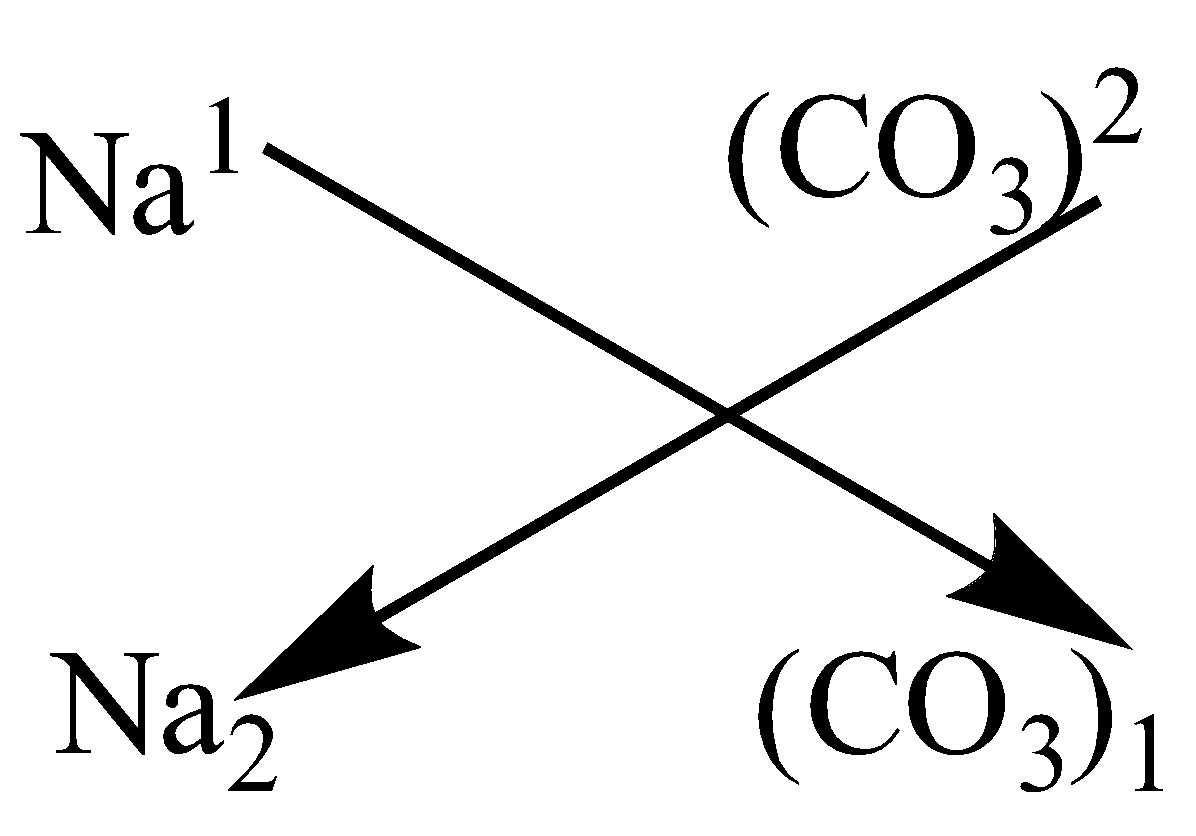
So, the charge is swapped and maintained accordingly. The chemical formula of sodium carbonate is \[N{{a}_{2}}(C{{O}_{3}})\].
Therefore, the correct option is C.
Additional information:
Sodium carbonate- disodium salt of carbonic acid. It exists in three forms of hydrates:
\[N{{a}_{2}}C{{O}_{3}}.\text{10}{{H}_{2}}O\](sodium carbonate decahydrate)
\[N{{a}_{2}}C{{O}_{3}}.7{{H}_{2}}O\](sodium carbonate heptahydrate)
\[N{{a}_{2}}C{{O}_{3}}.{{H}_{2}}O\](sodium carbonate monohydrate)
The given compound in the question is the anhydrous salt (salt without any water molecule). Another name for this anhydrous salt is calcined soda. It is manufactured in the last step of the Solvay process when sodium hydrogen carbonate is calculated.
Note: The chemical formula of an ionic compound or a covalent compound tells us the ratio of the elements present in it.
The valency of an atom or ion is the number of electrons it shares, loses or gains during a chemical reaction to attain stability that is the number of bonds it forms with the other atoms.
Complete step by step answer:
The valency of a complex ion is the same as the value of the charge of the ion.
Carbonate (\[C{{O}_{3}}^{2-}\]) has a charge of -2, so valency is 2. Valency of sodium (\[N{{a}^{+}}\]) is 1.
Hence, as per crossover rule, crossing valency over:

So, the charge is swapped and maintained accordingly. The chemical formula of sodium carbonate is \[N{{a}_{2}}(C{{O}_{3}})\].
Therefore, the correct option is C.
Additional information:
Sodium carbonate- disodium salt of carbonic acid. It exists in three forms of hydrates:
\[N{{a}_{2}}C{{O}_{3}}.\text{10}{{H}_{2}}O\](sodium carbonate decahydrate)
\[N{{a}_{2}}C{{O}_{3}}.7{{H}_{2}}O\](sodium carbonate heptahydrate)
\[N{{a}_{2}}C{{O}_{3}}.{{H}_{2}}O\](sodium carbonate monohydrate)
The given compound in the question is the anhydrous salt (salt without any water molecule). Another name for this anhydrous salt is calcined soda. It is manufactured in the last step of the Solvay process when sodium hydrogen carbonate is calculated.
Note: The chemical formula of an ionic compound or a covalent compound tells us the ratio of the elements present in it.
The valency of an atom or ion is the number of electrons it shares, loses or gains during a chemical reaction to attain stability that is the number of bonds it forms with the other atoms.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Explain the Treaty of Vienna of 1815 class 10 social science CBSE

What is the full form of CNG A Complete Natural Gas class 10 social science CBSE

In cricket, what is a "Yorker" designed to do?

What is the full form of POSCO class 10 social science CBSE

Define Potential, Developed, Stock and Reserved resources

What were the majoritarian measures taken in Sri Lanka class 10 social science CBSE




