$C{{H}_{3}}-CH=C{{H}_{2}}+HBr\to $
The major product formed is:
[A] $C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-Br$
[B] $C{{H}_{3}}-CH(Br)-C{{H}_{3}}$
[C] $C{{H}_{2}}Br-CH=C{{H}_{2}}$
[D] $C{{H}_{2}}=C=C{{H}_{2}}$
Answer
598.2k+ views
HINT: To answer this, you need to remember Markonikov’s addition. Here, the reaction will be an additional reaction. Two carbocations might be formed here but the secondary carbocation will be more stable and will give you the major product.
Complete step by step solution: Here, we can see that the compound given to us is an alkene named propene. It will react readily with hydrogen bromide and it will give us an alkyl bromide compound. Now, let us see the reaction to find out the major product.
Here, when we add HBr, it undergoes Markonikov’s addition giving us the product.
According to the Markovnikov’s Rule, bromine will be added to the most substituted carbon, giving us the major product by undergoing an additional reaction.
Now, let us see the mechanism of the addition.
We know that the H – Br bond is polarised due the electronegativity difference between them. Also, in the alkene the electron density is higher on the double bond.
The ${{H}^{+}}$ ion will attack on the propene carbocation and there is a possibility of formation of two carbocation - one primary, the other secondary. However, secondary carbocation is more stable so it will give us the major product. Then, the bromide ion reacts with the secondary carbocation formed and gives us an alkyl bromide.
We can show the mechanism as-
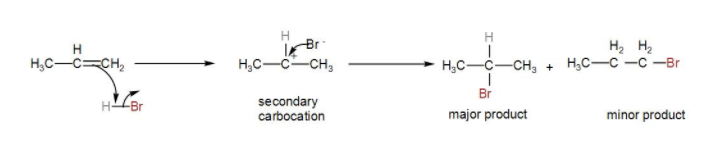
Therefore, the correct answer is option [B] $C{{H}_{3}}-CH(Br)-C{{H}_{3}}$
NOTE: It is important for us to remember that HBr shows peroxide effect i.e.in presence of HBr and peroxide, it changes the region selectivity and gives the opposite of the expected product. The product thus obtained is the Anti-Markovnikov product. However, hydrogen chloride and hydrogen iodide do not show a peroxide effect.
Complete step by step solution: Here, we can see that the compound given to us is an alkene named propene. It will react readily with hydrogen bromide and it will give us an alkyl bromide compound. Now, let us see the reaction to find out the major product.
Here, when we add HBr, it undergoes Markonikov’s addition giving us the product.
According to the Markovnikov’s Rule, bromine will be added to the most substituted carbon, giving us the major product by undergoing an additional reaction.
Now, let us see the mechanism of the addition.
We know that the H – Br bond is polarised due the electronegativity difference between them. Also, in the alkene the electron density is higher on the double bond.
The ${{H}^{+}}$ ion will attack on the propene carbocation and there is a possibility of formation of two carbocation - one primary, the other secondary. However, secondary carbocation is more stable so it will give us the major product. Then, the bromide ion reacts with the secondary carbocation formed and gives us an alkyl bromide.
We can show the mechanism as-

Therefore, the correct answer is option [B] $C{{H}_{3}}-CH(Br)-C{{H}_{3}}$
NOTE: It is important for us to remember that HBr shows peroxide effect i.e.in presence of HBr and peroxide, it changes the region selectivity and gives the opposite of the expected product. The product thus obtained is the Anti-Markovnikov product. However, hydrogen chloride and hydrogen iodide do not show a peroxide effect.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




