How can I calculate the osmolarity of dextrose?
Answer
559.8k+ views
Hint: Osmolarity is a measure of the concentration of a solution in terms of the total osmoles (osm) of solute particles present in one liter of a solution. The SI unit of osmolarity is osm/L.
An osmole can be defined as the number of moles of a solute that contribute to the osmotic pressure of a solution.
Complete answer:
The osmolarity of a solution can be given by the formula
\[osmolarity=\sum\limits_{i}{{{\varphi }_{i}}{{n}_{i}}{{C}_{i}}}\]
Here, the osmotic coefficient $\varphi $ gives the degree of non-ideality of a solution. When there is a 100% dissociation, $\varphi $=1.
n is the number of ions that are formed upon dissociation,
C is the molar concentration of the solute, and
i is the index representing the identity of a solute.
Now, osmolarity is used to measure the osmoles of the solute particles. It is dependent on the property of a substance to dissociate.
Compounds that dissociate in a solution do not have a 1:1 ratio between osmolarity and molarity.
For example, ionic compounds such as NaCl salt dissociates into the solution like
\[NaCl\to N{{a}^{+}}+O{{H}^{-}}\]
So, 1 mole of a solution of NaCl dissociates into 2 osmoles of solute particles.
On the other hand, compounds that do not dissociate, like non-ionic compounds, have a 1:1 ratio between osmolarity and molarity i.e., one mol of solution forms 1 osmole.
We know that dextrose is a simple sugar and is chemically identical to D-glucose. It has the molecular formula ${{C}_{6}}{{H}_{12}}{{O}_{6}}$.
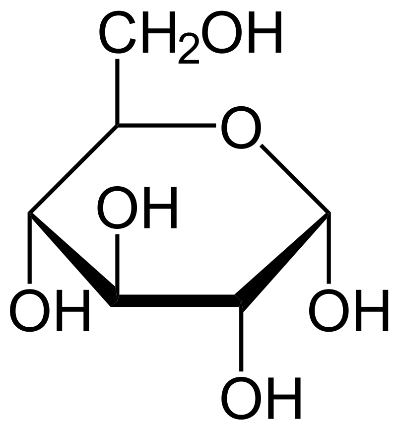
Since glucose is a non-ionic compound, it does not dissociate in an aqueous solution. Hence 1 mol solution of glucose forms 1osmol.
Similarly, 1 mol/L solution of dextrose is 1 osmol/L.
Note:
It must be noted that there can be multiple compounds that contribute to the osmolarity of a solution. It means that a N osm solution might consist of a moles of compound A, b moles of compound B, c moles of compound C, etc., and the sum of all the moles will be equal to n.
\[a+b+c+.........=N\]
An osmole can be defined as the number of moles of a solute that contribute to the osmotic pressure of a solution.
Complete answer:
The osmolarity of a solution can be given by the formula
\[osmolarity=\sum\limits_{i}{{{\varphi }_{i}}{{n}_{i}}{{C}_{i}}}\]
Here, the osmotic coefficient $\varphi $ gives the degree of non-ideality of a solution. When there is a 100% dissociation, $\varphi $=1.
n is the number of ions that are formed upon dissociation,
C is the molar concentration of the solute, and
i is the index representing the identity of a solute.
Now, osmolarity is used to measure the osmoles of the solute particles. It is dependent on the property of a substance to dissociate.
Compounds that dissociate in a solution do not have a 1:1 ratio between osmolarity and molarity.
For example, ionic compounds such as NaCl salt dissociates into the solution like
\[NaCl\to N{{a}^{+}}+O{{H}^{-}}\]
So, 1 mole of a solution of NaCl dissociates into 2 osmoles of solute particles.
On the other hand, compounds that do not dissociate, like non-ionic compounds, have a 1:1 ratio between osmolarity and molarity i.e., one mol of solution forms 1 osmole.
We know that dextrose is a simple sugar and is chemically identical to D-glucose. It has the molecular formula ${{C}_{6}}{{H}_{12}}{{O}_{6}}$.

Since glucose is a non-ionic compound, it does not dissociate in an aqueous solution. Hence 1 mol solution of glucose forms 1osmol.
Similarly, 1 mol/L solution of dextrose is 1 osmol/L.
Note:
It must be noted that there can be multiple compounds that contribute to the osmolarity of a solution. It means that a N osm solution might consist of a moles of compound A, b moles of compound B, c moles of compound C, etc., and the sum of all the moles will be equal to n.
\[a+b+c+.........=N\]
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




