How do you calculate the dipole moment of water?
Answer
598.8k+ views
Hint: Dipole moment is a vector quantity means it has magnitude and direction. Dipole moment is a measure of polarity of chemical bonds present in between two atoms in a particular molecule. In dipole moment the separation of negative and positive charge takes place.
Complete answer:
- In the question they asked how we can calculate the dipole moment of water.
- There is a formula to calculate the dipole moment and it is as follows.
\[\text{Dipole moment (}\mu \text{) = charge (Q) }\times \text{ distance of separation (r)}\]
- Now coming to the calculation of the dipole moment of the water molecule.
- We know that the electrons are localized around the oxygen atom in the water molecule.
- The localization of electrons is due to the high electronegativity of the central oxygen atom in water molecules.
- Due to the presence of lone pairs of electrons on the oxygen molecule the shape of the molecule is going to be a bent shape.
- The shape and charge separation in the water can be seen in the following picture.
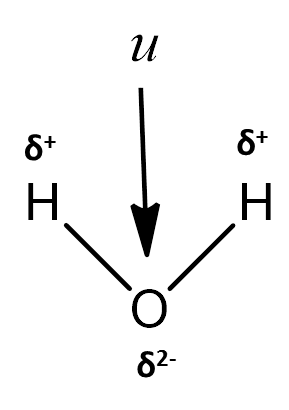
- The bond angle which is present in water is 104.5$^{o}$ .
- The individual bond moment of each hydrogen-oxygen bond is 1.5 D.
- There are two hydrogen in water molecules, the two hydrogen creates their individual dipole moments.
- We have to calculate the individual dipole moments and later we have to do the sum to get the dipole moment of the water molecule.
- The dipole moment caused by the left hydrogen in water molecule = $1.5D\times \cos ({{52.2388}^{o}})=15D\times 0.612 = 0.9187$
(Here, ${{52.2388}^{o}}=\dfrac{{{104.5}^{o}}}{2}$ )
- The dipole moment caused by the right hydrogen in water molecule = $1.5D\times \cos ({{52.2388}^{o}})=15D\times 0.612 = 0.9187$
(Here, ${{52.2388}^{o}}=\dfrac{{{104.5}^{o}}}{2}$ )
Therefore the net dipole moment of the water molecule = $0.9187 + 0.9187 = 1.837 D$.
Note: The unit to measure dipole moment of the molecules is D (Debye). The dipole moment for linear molecules like beryllium difluoride are 0. Because the dipole moment caused by individual fluorine atoms is zero.
Complete answer:
- In the question they asked how we can calculate the dipole moment of water.
- There is a formula to calculate the dipole moment and it is as follows.
\[\text{Dipole moment (}\mu \text{) = charge (Q) }\times \text{ distance of separation (r)}\]
- Now coming to the calculation of the dipole moment of the water molecule.
- We know that the electrons are localized around the oxygen atom in the water molecule.
- The localization of electrons is due to the high electronegativity of the central oxygen atom in water molecules.
- Due to the presence of lone pairs of electrons on the oxygen molecule the shape of the molecule is going to be a bent shape.
- The shape and charge separation in the water can be seen in the following picture.

- The bond angle which is present in water is 104.5$^{o}$ .
- The individual bond moment of each hydrogen-oxygen bond is 1.5 D.
- There are two hydrogen in water molecules, the two hydrogen creates their individual dipole moments.
- We have to calculate the individual dipole moments and later we have to do the sum to get the dipole moment of the water molecule.
- The dipole moment caused by the left hydrogen in water molecule = $1.5D\times \cos ({{52.2388}^{o}})=15D\times 0.612 = 0.9187$
(Here, ${{52.2388}^{o}}=\dfrac{{{104.5}^{o}}}{2}$ )
- The dipole moment caused by the right hydrogen in water molecule = $1.5D\times \cos ({{52.2388}^{o}})=15D\times 0.612 = 0.9187$
(Here, ${{52.2388}^{o}}=\dfrac{{{104.5}^{o}}}{2}$ )
Therefore the net dipole moment of the water molecule = $0.9187 + 0.9187 = 1.837 D$.
Note: The unit to measure dipole moment of the molecules is D (Debye). The dipole moment for linear molecules like beryllium difluoride are 0. Because the dipole moment caused by individual fluorine atoms is zero.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

What are hard magnetic materials class 11 physics CBSE




