Aniline is treated with $B{{r}_{2}}$ water at room temperature to give the following product:
(a)
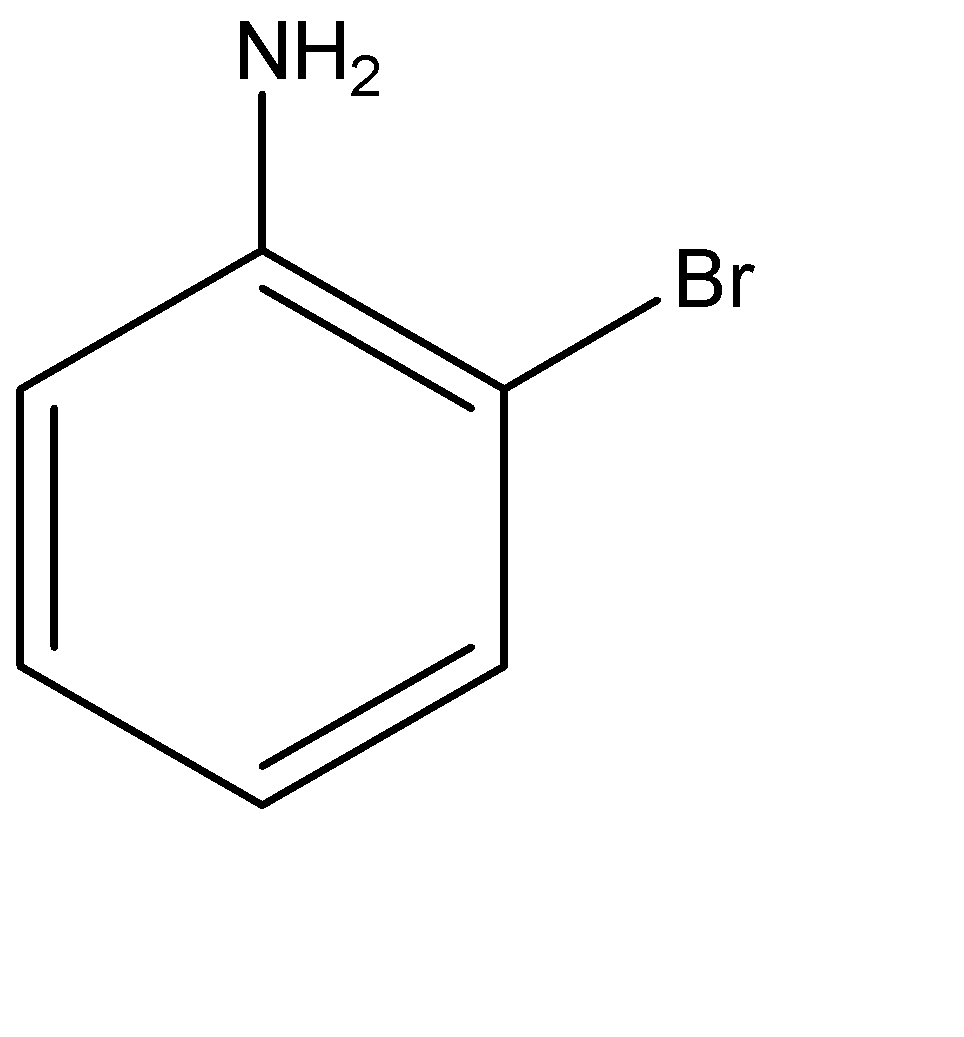
(b)
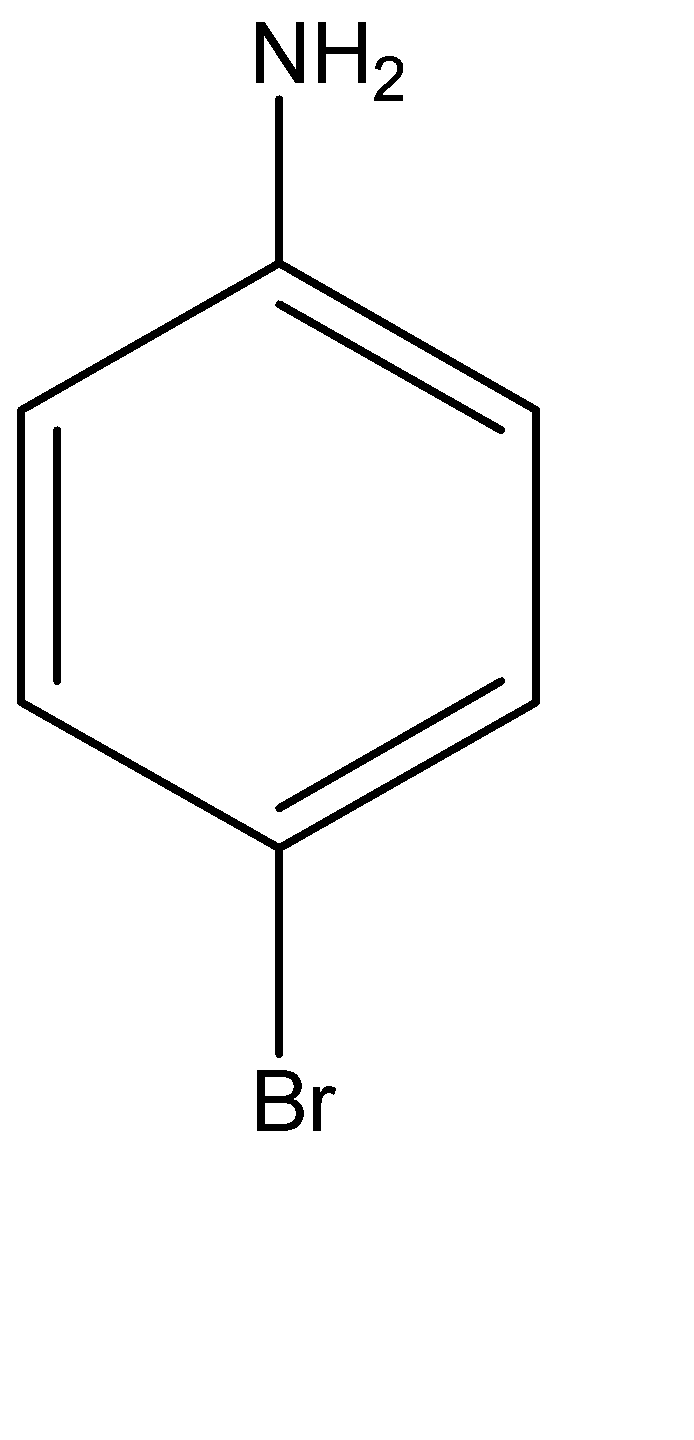
(c)
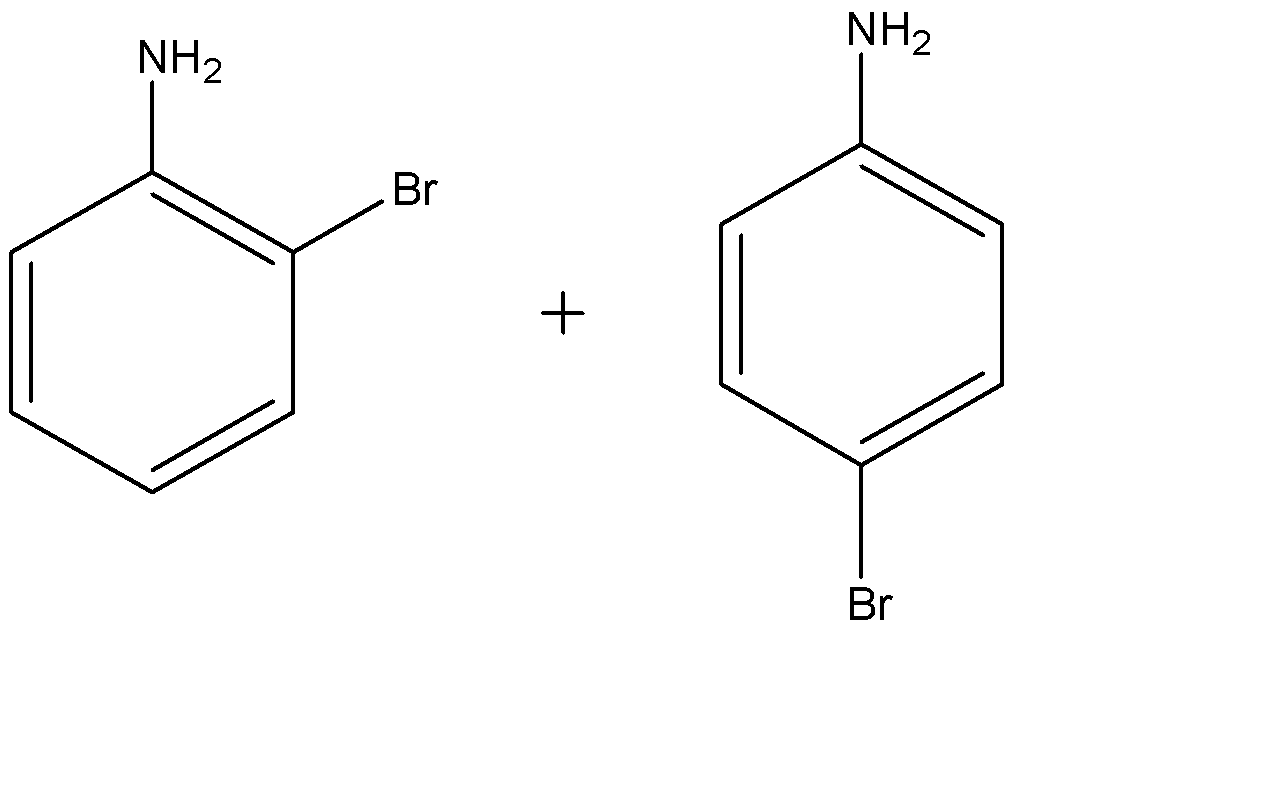
(d)
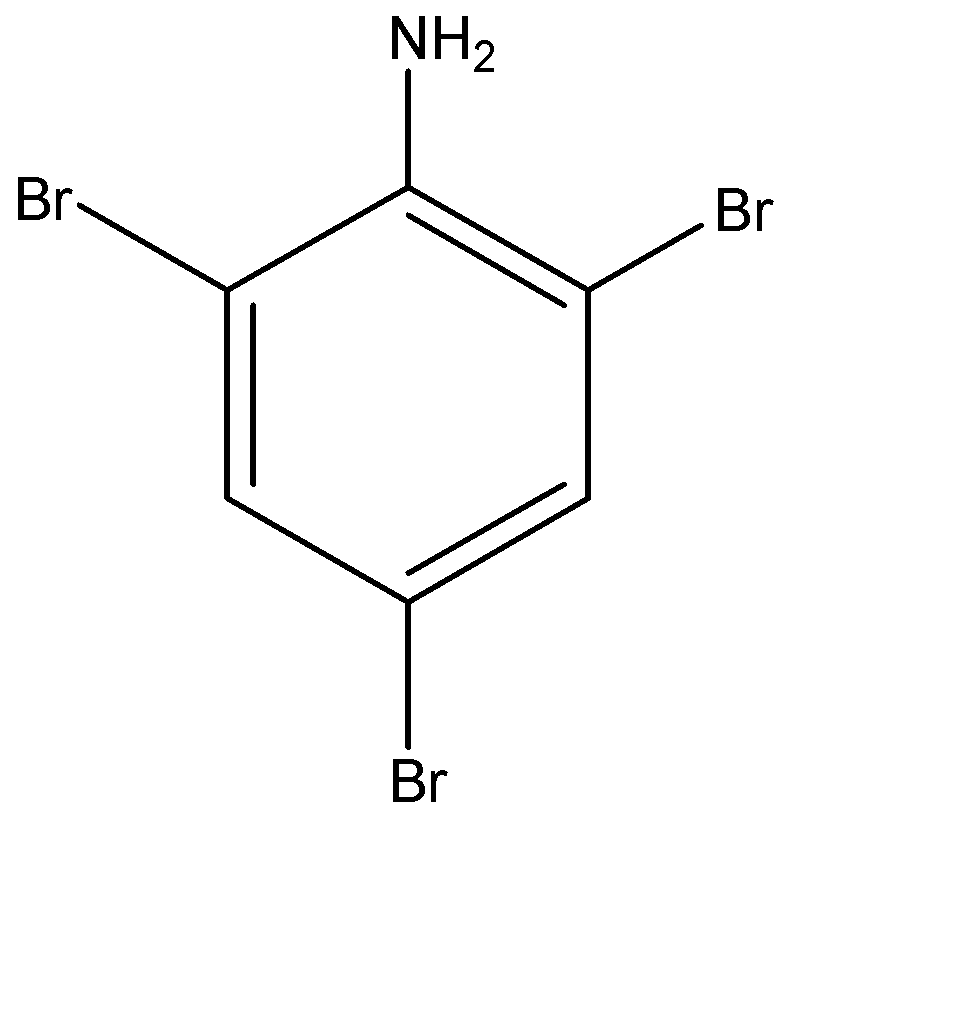




Answer
552.3k+ views
Hint: As we know that aniline is an organic compound that consists of a phenyl group attached to an amino group. It is the simplest aromatic amine available. It is industrially very significant and used as a starting reaction in organic synthesis of various compounds. So here we are to tell the compound formed when aniline is treated with Bromine water at room temperature.
Complete answer:
Let us discuss about reactive nature of aniline followed by the reaction as follows:-
Just like phenols, aniline does give electrophilic substitution reaction. Due to its high reactivity, it can also be categorized as an enamine. This is because aniline has a good electron donating ability to the electrophilic species. This can be seen in the reaction of aniline with bromine water at room temperature as follows:-
-Bromination
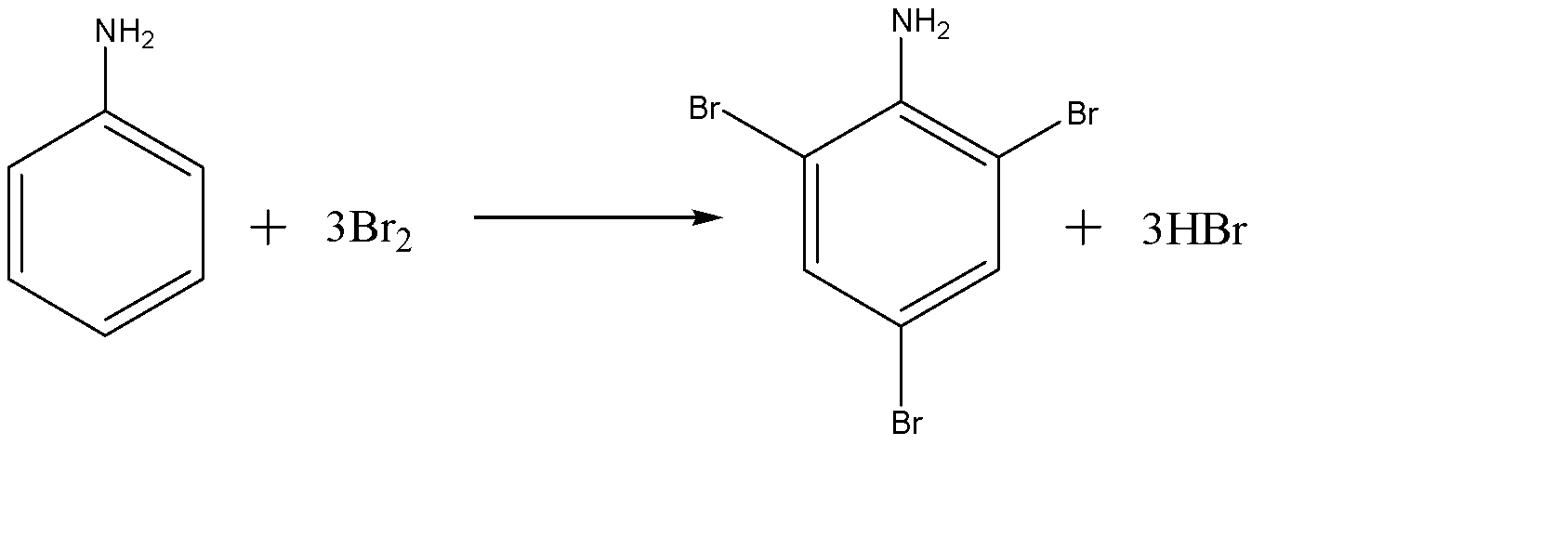
The benzene ring of aniline is so highly activated due to strong mesomeric effect amino group that when it is treated with bromine water ($B{{r}_{2}}$ ) at room temperature, electrophilic substitution takes place at all para and ortho positions which results in the formation of 2,4,6-tribromoaniline.
-The formation of 2,4,6-tribromoaniline can be seen because a white colour precipitate is formed in the solution and also the yellowish orange colour of bromine water also disappears at the same time.
-Therefore we can say that, aniline is treated with $B{{r}_{2}}$ water at room temperature to give following product:-
(d)
Note:
-To obtain a monosubstituted product, we need to decrease the effect of the amino group by acetylating this group before the substitution reaction.
-Also study and learn all types of reaction shown by aniline as it is an important reactant in most of the organic synthesis.
Complete answer:
Let us discuss about reactive nature of aniline followed by the reaction as follows:-
Just like phenols, aniline does give electrophilic substitution reaction. Due to its high reactivity, it can also be categorized as an enamine. This is because aniline has a good electron donating ability to the electrophilic species. This can be seen in the reaction of aniline with bromine water at room temperature as follows:-
-Bromination

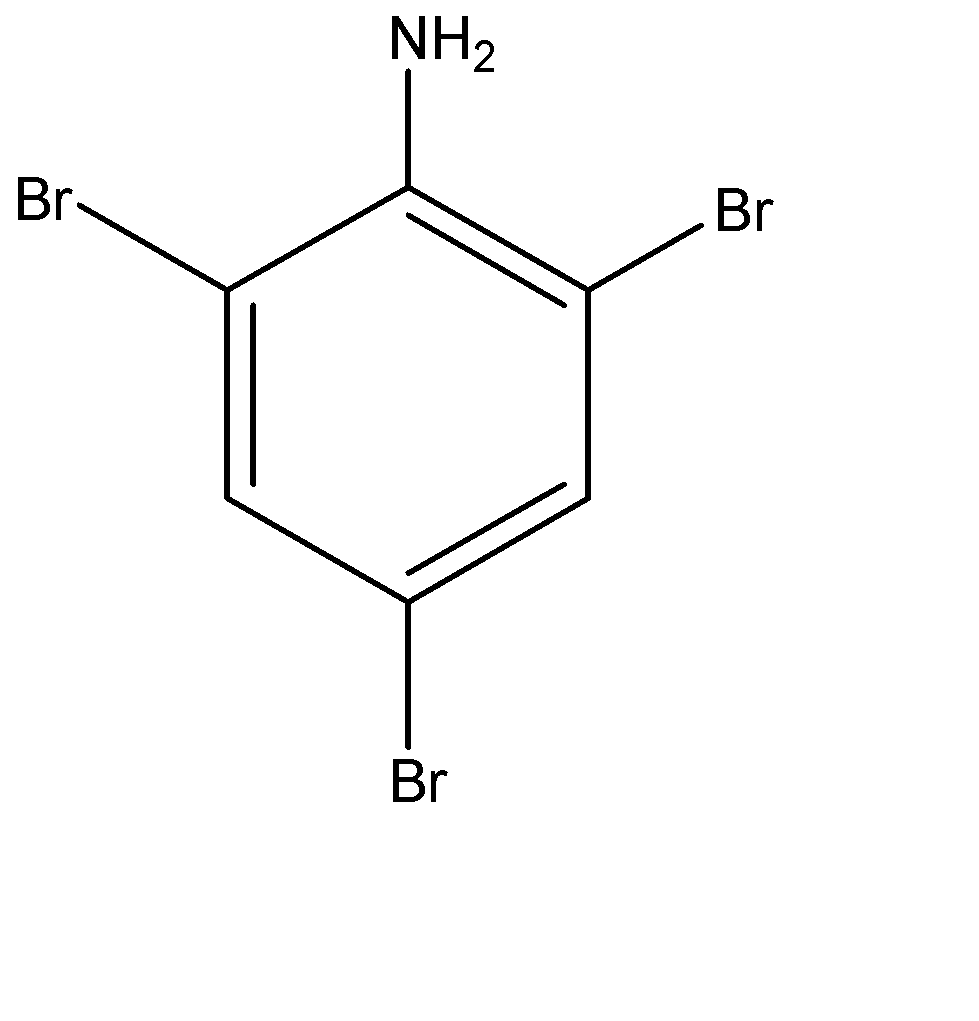
The benzene ring of aniline is so highly activated due to strong mesomeric effect amino group that when it is treated with bromine water ($B{{r}_{2}}$ ) at room temperature, electrophilic substitution takes place at all para and ortho positions which results in the formation of 2,4,6-tribromoaniline.
-The formation of 2,4,6-tribromoaniline can be seen because a white colour precipitate is formed in the solution and also the yellowish orange colour of bromine water also disappears at the same time.
-Therefore we can say that, aniline is treated with $B{{r}_{2}}$ water at room temperature to give following product:-
(d)

Note:
-To obtain a monosubstituted product, we need to decrease the effect of the amino group by acetylating this group before the substitution reaction.
-Also study and learn all types of reaction shown by aniline as it is an important reactant in most of the organic synthesis.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Find the value of the expression given below sin 30circ class 11 maths CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE

What are hard magnetic materials class 11 physics CBSE




