(a) Write the electron dot structure of calcium and sulphur.
(b) Show the formation of CaS by transfer of electrons.
Answer
547.5k+ views
Hint: Lewis dot structures or electron dot structures show the bonding between atoms in a molecule. Electrons are represented as dots in the Lewis structures. To draw the electron dot structure of atoms, we must know the atomic number of them. The total number of electrons represented in the Lewis dot structure will be equal to the total number of valence electrons in an atom.
Complete answer:
(a)Atomic number of calcium and sulphur are $20\,and\,16$ respectively.
Calcium is an alkaline earth metal and belongs to group $2$ & period $4$ . The electronic configuration is $1{s^2}2{s^2}2{p^6}3{s^2}3{p^6}4{s^2}$ . The electron distribution in shells is given as $2,8,8,2$ . Therefore the total number of valence electrons present are $2$ . The electron dot structure of Ca is:

Sulphur is a chalcogen belonging to group $16$ & period $3$ of the periodic table. The electronic configuration is $1{s^2}2{s^2}2{p^6}3{s^2}3{p^4}$ and the electron configuration of shells is given by $2,8,6$ . Thus, total number of valence electrons are $6$ .The electron dot structure of S is:
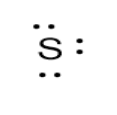
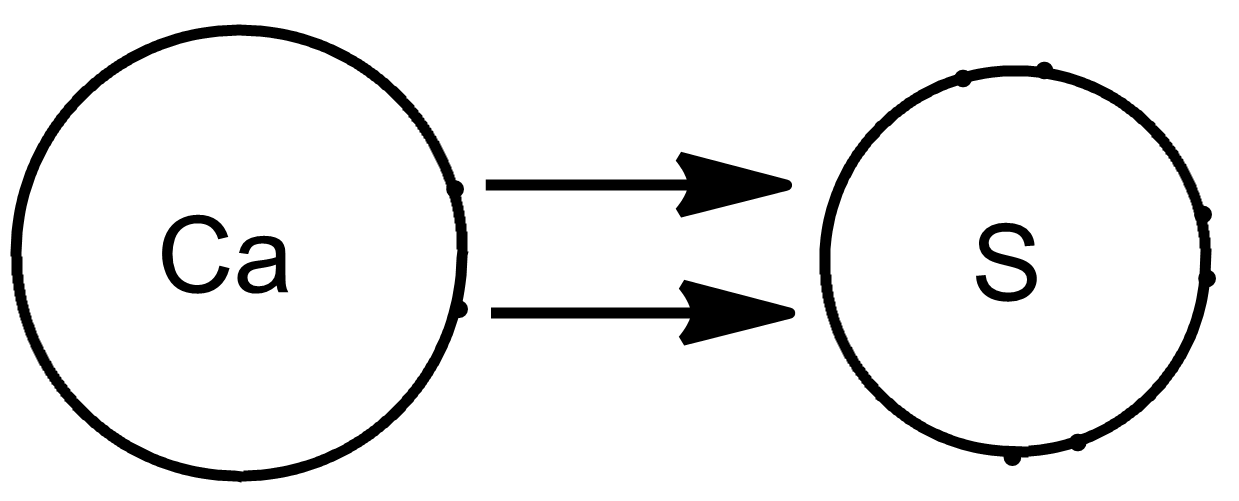
(b)Calcium has to donate its two valence electrons while sulphur has to accept a pair of electrons on its valence shell in order to attain the nearest inert gas configuration and become stable. Therefore, the transfer of electrons occur as:
$Ca\, \to \,C{a^{2 + }}\, + \,2\mathop e\limits^ -$
$ S\, + \,2\mathop e\limits^ - \to \,{S^{2 - }}$
The transfer of electrons is shown by an arrow.
Thus, the ionic compound thus formed is Calcium sulphide is shown below.
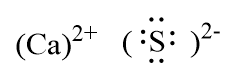
Note:
It has to be taken note that the electrons represented in a Lewis dot representation are the valence electrons of an atom i.e., the electrons present in the outermost orbital or valence shell of the atom. Non valence electrons are not depicted in this representation. Such structures can be used to represent covalently bonded and coordination compounds as well.
Complete answer:
(a)Atomic number of calcium and sulphur are $20\,and\,16$ respectively.
Calcium is an alkaline earth metal and belongs to group $2$ & period $4$ . The electronic configuration is $1{s^2}2{s^2}2{p^6}3{s^2}3{p^6}4{s^2}$ . The electron distribution in shells is given as $2,8,8,2$ . Therefore the total number of valence electrons present are $2$ . The electron dot structure of Ca is:

Sulphur is a chalcogen belonging to group $16$ & period $3$ of the periodic table. The electronic configuration is $1{s^2}2{s^2}2{p^6}3{s^2}3{p^4}$ and the electron configuration of shells is given by $2,8,6$ . Thus, total number of valence electrons are $6$ .The electron dot structure of S is:
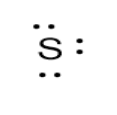
(b)Calcium has to donate its two valence electrons while sulphur has to accept a pair of electrons on its valence shell in order to attain the nearest inert gas configuration and become stable. Therefore, the transfer of electrons occur as:
$Ca\, \to \,C{a^{2 + }}\, + \,2\mathop e\limits^ -$
$ S\, + \,2\mathop e\limits^ - \to \,{S^{2 - }}$
The transfer of electrons is shown by an arrow.

Thus, the ionic compound thus formed is Calcium sulphide is shown below.
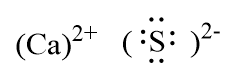
Note:
It has to be taken note that the electrons represented in a Lewis dot representation are the valence electrons of an atom i.e., the electrons present in the outermost orbital or valence shell of the atom. Non valence electrons are not depicted in this representation. Such structures can be used to represent covalently bonded and coordination compounds as well.
Recently Updated Pages
Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Master Class 9 Social Science: Engaging Questions & Answers for Success

Class 9 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

10 examples of friction in our daily life

Name the Largest and the Smallest Cell in the Human Body ?




