
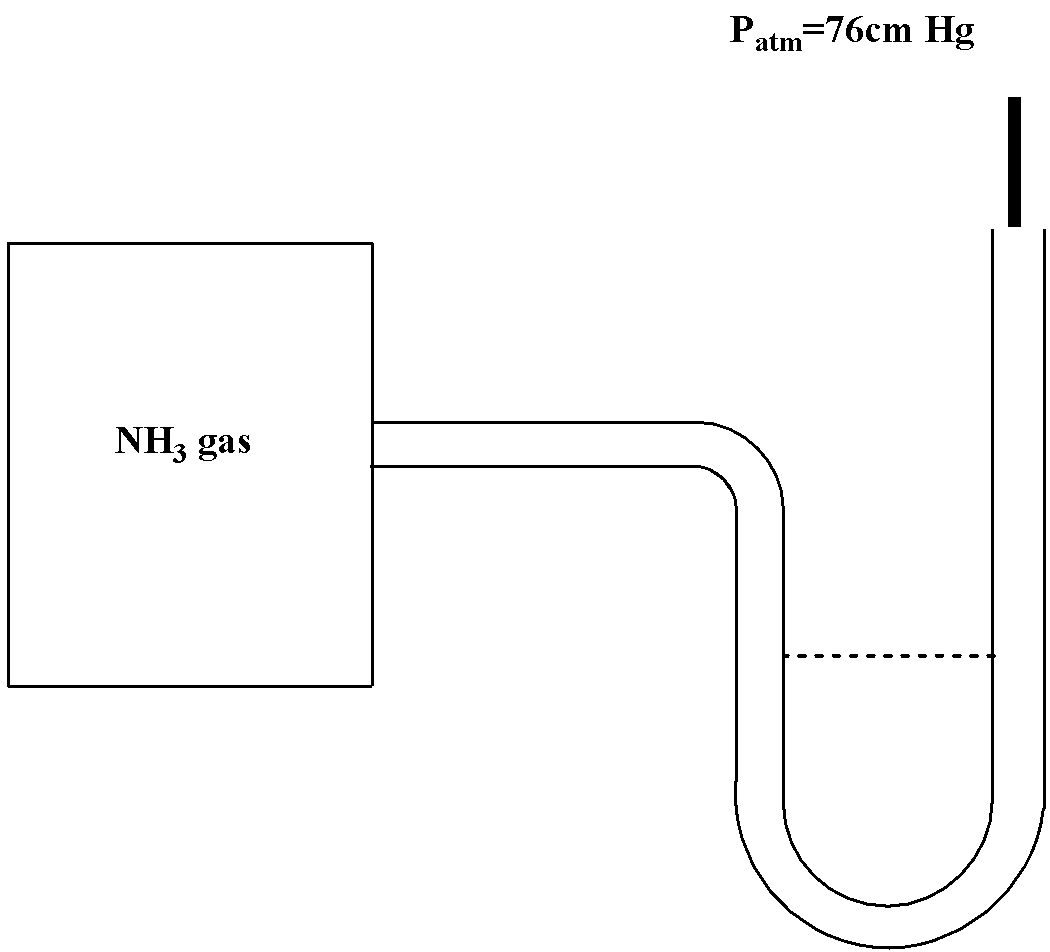
A manometer attached to a flask containing ammonia gas has no difference in mercury level initially as shown in diagram. After sparking into the flask, ammonia is partially dissociated as $2N{H_3}(g) \to {N_2}(g) + 3{H_2}(g)$ now it have difference of 6 cm in mercury level in two columns, what is partial pressure of ${H_2}(g)$ at equilibrium?
(A) 6 mm Hg
(B) 18 mm Hg
(C) 27 mm Hg
(D) None of these

Answer
553.8k+ views
Hint: According to ideal gas law, partial pressure is inversely proportional to volume. It is directly proportional to the number of moles and temperature. Then the pressure at initial and pressure at equilibrium will be found.
Complete step by step answer:
We have been given the question that a manometer attached to the flask contains ammonia gas.
Then we have been given that ammonia is partially dissociated to nitrogen gas and hydrogen gas as follows
$2N{H_3}(g) \to {N_2}(g) + 3{H_2}(g)$
It is given that ammonia is at $76$ cm Hg pressure. Therefore the initial pressure will be 76 cm Hg
At equilibrium, the pressure will be $76 - 2x,x,3x$ respectively. So the total pressure will be written as $76 - 2x + x + 3x = 76 + 2x$
So the total pressure at equilibrium is $76 + 2x$
We can observe from here that the increase in pressure is $76 - (76 + 2x) = 2x$
It is given that difference in mercury level is six cm
$ \Rightarrow 2x = 6$
$ \Rightarrow x = 3$ cm
So the partial pressure of hydrogen gas $ = 3x$
$ \Rightarrow 3 \times 3 = 9\;cm\;Hg$
So the partial pressure of hydrogen is $9\;cm\;Hg$
So, the correct answer is Option D.
Additional information:
The difference between partial pressure and vapor pressure is that partial pressure is the pressure exerted by the gas individually in the mixture. While vapor pressure is the pressure exerted by the liquid at the thermodynamic equilibrium.
Note: The partial pressure of an individual gas is equal to the total pressure multiplied by the mole fraction of that gas.
The partial pressure of gas $ = $ Total pressure $ \times $ mole fraction of gas
$ \Rightarrow P = {P_T}{X_A}$
Also, the total pressure of an ideal gas mixture is the sum of the partial pressure of the gases in the mixture. The pressure exerted by vapors of liquid is called vapor pressure.
Complete step by step answer:
We have been given the question that a manometer attached to the flask contains ammonia gas.
Then we have been given that ammonia is partially dissociated to nitrogen gas and hydrogen gas as follows
$2N{H_3}(g) \to {N_2}(g) + 3{H_2}(g)$
It is given that ammonia is at $76$ cm Hg pressure. Therefore the initial pressure will be 76 cm Hg
At equilibrium, the pressure will be $76 - 2x,x,3x$ respectively. So the total pressure will be written as $76 - 2x + x + 3x = 76 + 2x$
So the total pressure at equilibrium is $76 + 2x$
We can observe from here that the increase in pressure is $76 - (76 + 2x) = 2x$
It is given that difference in mercury level is six cm
$ \Rightarrow 2x = 6$
$ \Rightarrow x = 3$ cm
So the partial pressure of hydrogen gas $ = 3x$
$ \Rightarrow 3 \times 3 = 9\;cm\;Hg$
So the partial pressure of hydrogen is $9\;cm\;Hg$
So, the correct answer is Option D.
Additional information:
The difference between partial pressure and vapor pressure is that partial pressure is the pressure exerted by the gas individually in the mixture. While vapor pressure is the pressure exerted by the liquid at the thermodynamic equilibrium.
Note: The partial pressure of an individual gas is equal to the total pressure multiplied by the mole fraction of that gas.
The partial pressure of gas $ = $ Total pressure $ \times $ mole fraction of gas
$ \Rightarrow P = {P_T}{X_A}$
Also, the total pressure of an ideal gas mixture is the sum of the partial pressure of the gases in the mixture. The pressure exerted by vapors of liquid is called vapor pressure.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

Explain zero factorial class 11 maths CBSE




