What is Z in the following reaction sequence?
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\xrightarrow[(ii){{H}_{3}}P{{O}_{2}}+{{H}_{2}}O(iii)CO,HCl;anhydrousAlC{{l}_{3}}/CuCl]{(i)NaN{{O}_{2}}+HCl/273K}Z\]
a.) \[{{C}_{6}}{{H}_{5}}C{{O}_{2}}H\]
b.) \[{{C}_{6}}{{H}_{5}}OH\]
c.) \[{{C}_{6}}{{H}_{5}}CHO\]
d.) \[{{C}_{6}}{{H}_{6}}\]
Answer
615.6k+ views
Hint: A lot of uses are there with aniline. By using aniline we can prepare benzene diazonium chloride. Benzene diazonium chloride is the starting precursor to prepare so many compounds. The compound formed at the end of this reaction contains a carbonyl group and it gives a positive test result for tollens test and schiff's test but it gives a negative test result for the Fehling’s test.
Complete step by step answer:
The reaction given in the question contains three steps to prepare Z.
Step-1: In the first step aniline reacts with Sodium nitrate (\[NaN{{O}_{2}}\]) and hydrochloric acid (HCl) at 273K.
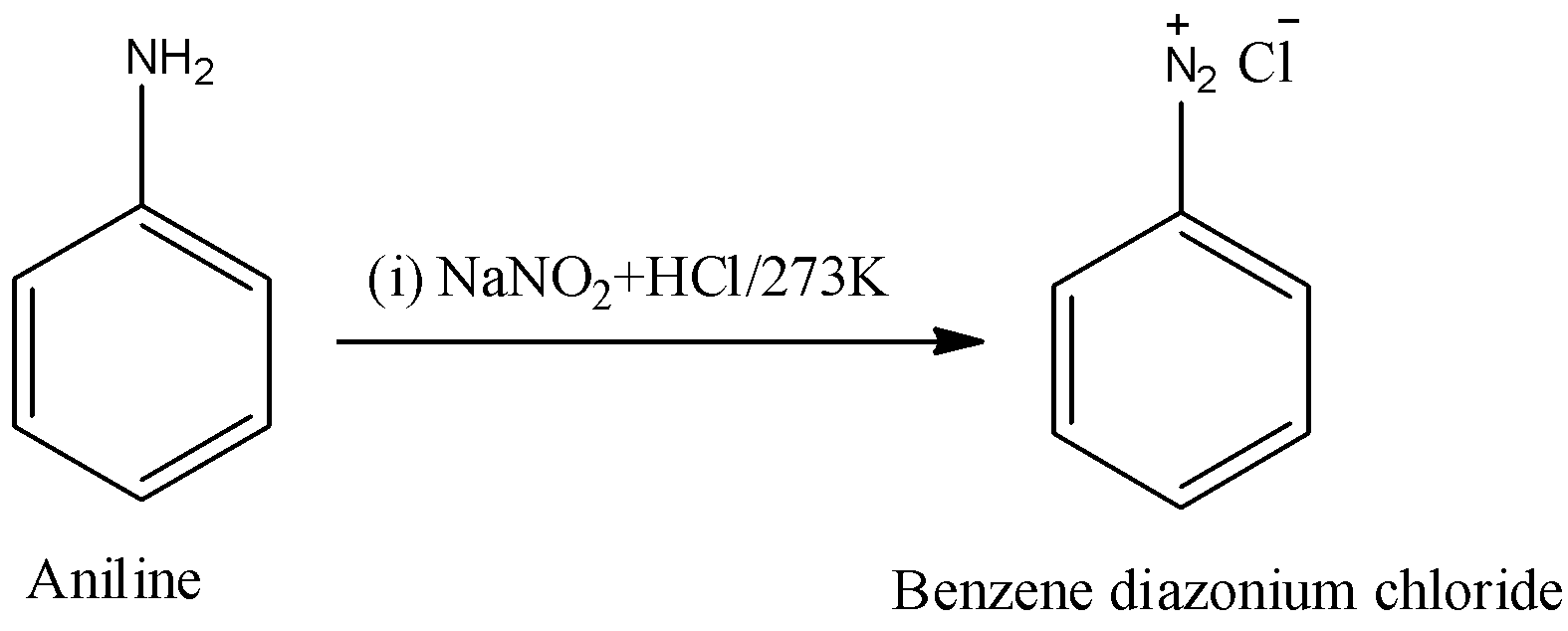
In the above reaction aniline is going to convert in to benzene diazonium chloride in the presence of Sodium nitrate (\[NaN{{O}_{2}}\]) and hydrochloric acid (HCl) at 273 K.
Step-2: The benzene diazonium chloride reacts with hypophosphorous acid and water.
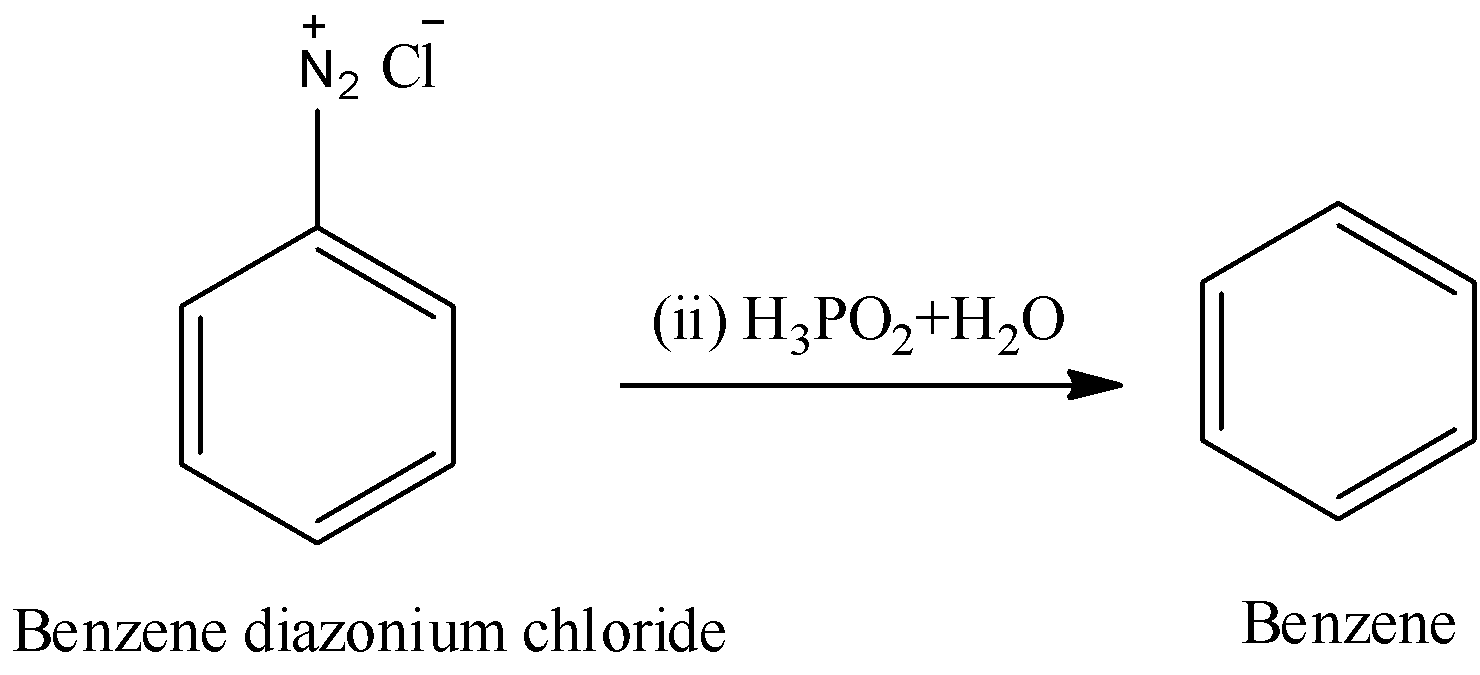
In the above reaction benzene diazonium chloride reacts with hypo phosphorous acid (\[{{H}_{3}}P{{O}_{2}}\]) and water and forms benzene as a product.
Step-3: In the third step benzene reacts with CO, HCl; anhydrous \[AlC{{l}_{3}}\]/CuCl.
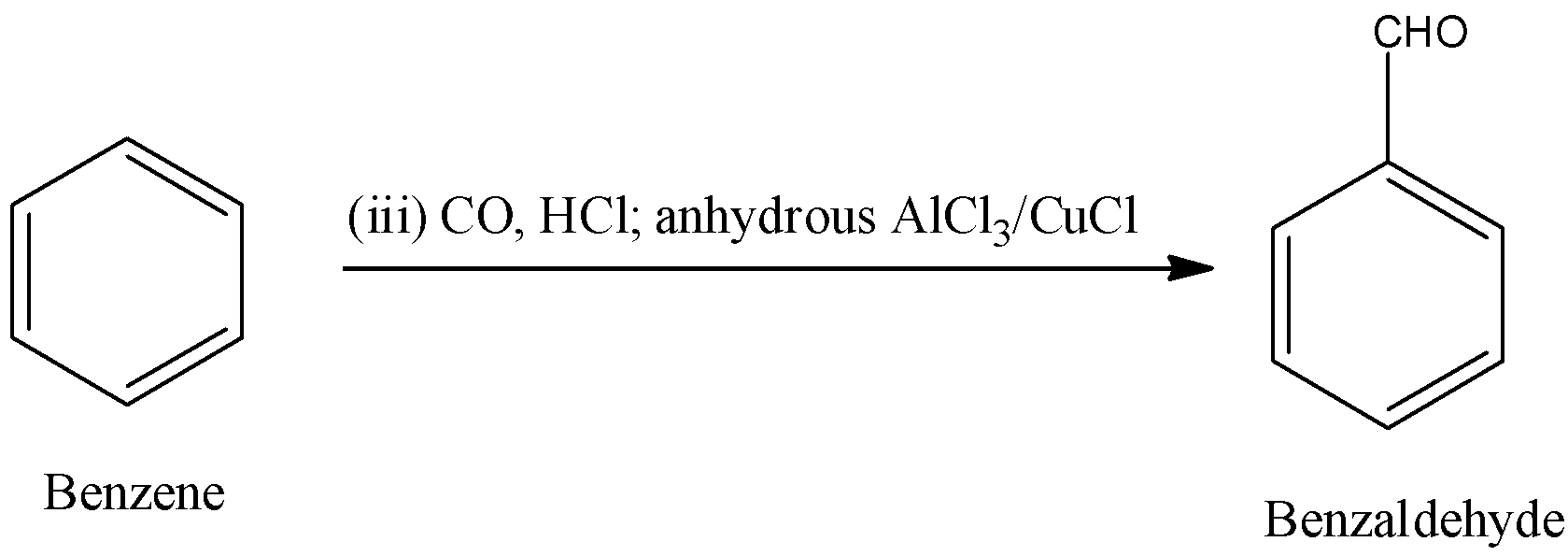
Benzene reacts with CO, HCl; anhydrous \[AlC{{l}_{3}}\]/CuCl and forms benzaldehyde as the product.
Therefore, we can write the overall reaction as follows.
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\xrightarrow[(ii){{H}_{3}}P{{O}_{2}}+{{H}_{2}}O(iii)CO,HCl;anhydrousAlC{{l}_{3}}/CuCl]{(i)NaN{{O}_{2}}+HCl/273K}{{C}_{6}}{{H}_{5}}CHO\]
Therefore the product Z is Benzaldehyde.
So, the correct answer is “Option C”.
Additional Information:
IUPAC name of benzaldehyde is benzenecarbaldehyde.
It is sometimes called benzenedicarboxaldehyde or benzoic aldehyde.
Benzaldehyde is miscible with ether, and alcohol.
Beekeepers usually apply Benzaldehyde as a bee repellent.
Benzaldehyde is commonly engaged to give almond flavor to foods.
Benzaldehyde is occasionally used in cosmetics products.
Note: By using Sandmeyer reaction we can prepare Chlorobenzene from benzene diazonium chloride.
.
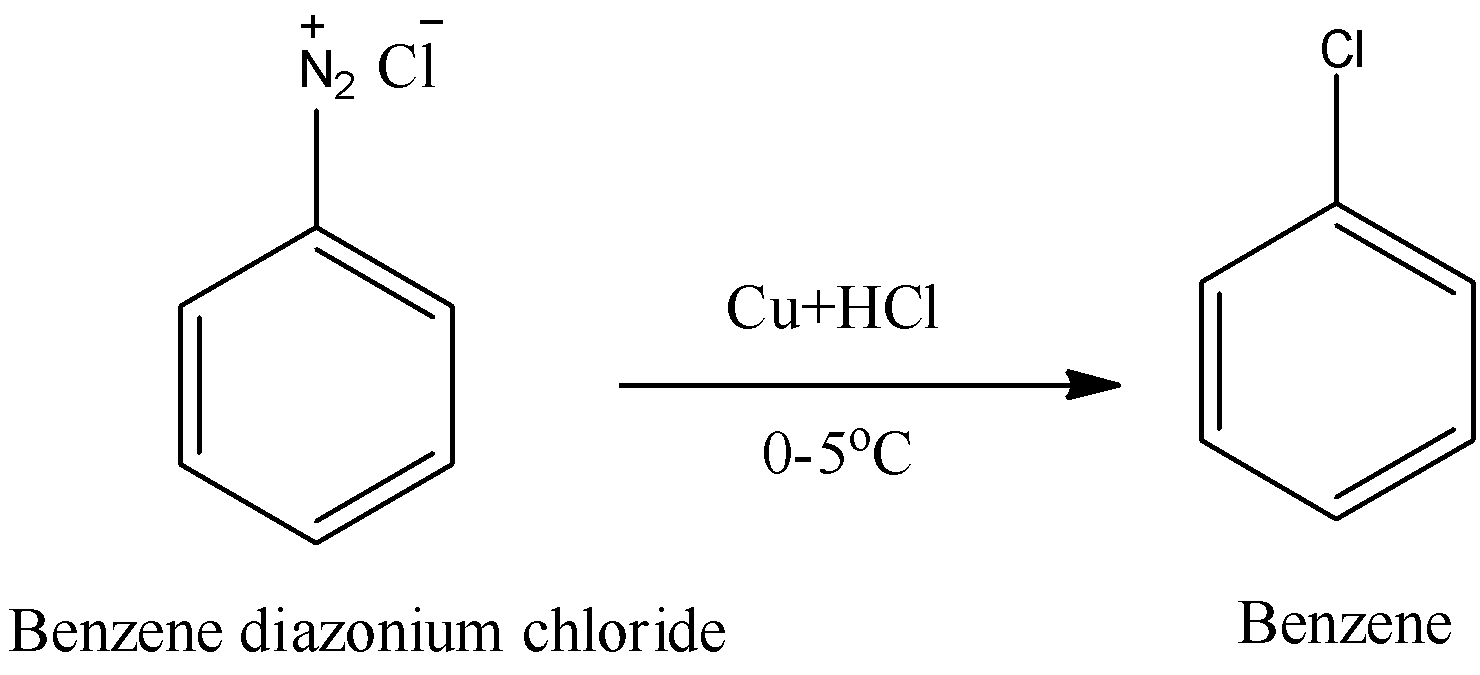
In step-2, Hypo phosphorous acid (\[{{H}_{3}}P{{O}_{2}}\]) gets oxidized to\[{{H}_{3}}P{{O}_{3}}\] and benzene is going to liberate from benzene diazonium chloride.
Complete step by step answer:
The reaction given in the question contains three steps to prepare Z.
Step-1: In the first step aniline reacts with Sodium nitrate (\[NaN{{O}_{2}}\]) and hydrochloric acid (HCl) at 273K.

In the above reaction aniline is going to convert in to benzene diazonium chloride in the presence of Sodium nitrate (\[NaN{{O}_{2}}\]) and hydrochloric acid (HCl) at 273 K.
Step-2: The benzene diazonium chloride reacts with hypophosphorous acid and water.

In the above reaction benzene diazonium chloride reacts with hypo phosphorous acid (\[{{H}_{3}}P{{O}_{2}}\]) and water and forms benzene as a product.
Step-3: In the third step benzene reacts with CO, HCl; anhydrous \[AlC{{l}_{3}}\]/CuCl.

Benzene reacts with CO, HCl; anhydrous \[AlC{{l}_{3}}\]/CuCl and forms benzaldehyde as the product.
Therefore, we can write the overall reaction as follows.
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\xrightarrow[(ii){{H}_{3}}P{{O}_{2}}+{{H}_{2}}O(iii)CO,HCl;anhydrousAlC{{l}_{3}}/CuCl]{(i)NaN{{O}_{2}}+HCl/273K}{{C}_{6}}{{H}_{5}}CHO\]
Therefore the product Z is Benzaldehyde.
So, the correct answer is “Option C”.
Additional Information:
IUPAC name of benzaldehyde is benzenecarbaldehyde.
It is sometimes called benzenedicarboxaldehyde or benzoic aldehyde.
Benzaldehyde is miscible with ether, and alcohol.
Beekeepers usually apply Benzaldehyde as a bee repellent.
Benzaldehyde is commonly engaged to give almond flavor to foods.
Benzaldehyde is occasionally used in cosmetics products.
Note: By using Sandmeyer reaction we can prepare Chlorobenzene from benzene diazonium chloride.
.

In step-2, Hypo phosphorous acid (\[{{H}_{3}}P{{O}_{2}}\]) gets oxidized to\[{{H}_{3}}P{{O}_{3}}\] and benzene is going to liberate from benzene diazonium chloride.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




