Write the structure of two chair conformation of 1- tert-butyl-2-methylcyclohexane. Which conformation is stable? Explain your answer.
Answer
615.3k+ views
Hint: Chair conformation is the most accurate representation to show how the cyclohexane rings are actually oriented in a space. It tells appropriately about the angle between the carbon atoms in a ring and also tells about the position of the group on each carbon atom present in a cyclohexane ring.
Complete step by step answer:
The structure of 1-tert-butyl-2- methylcyclohexane is given as below:
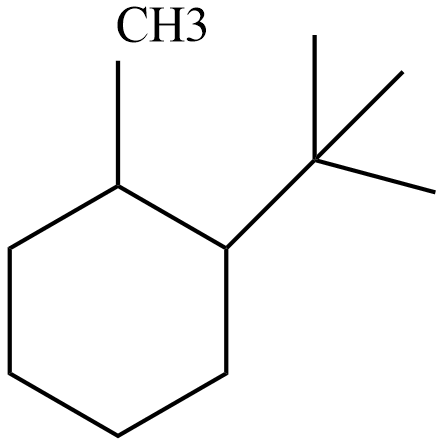
Now we will draw the two structure of chair conformation of 1-tert-butyl-2- methylcyclohexane as:

Figure 1.
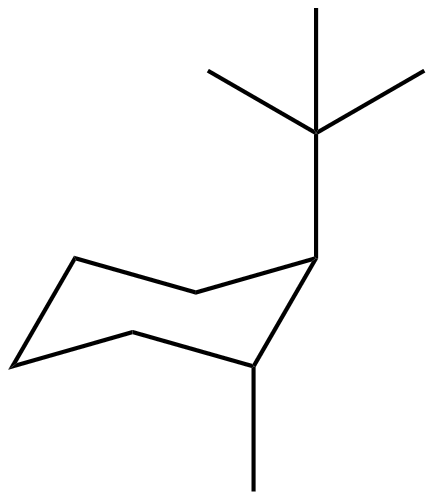
Figure 2.
The most stable chair conformation structure of 1-tert-butyl-2- methylcyclohexane is that chair conformation which is described by figure 1. as compared to that chair configuration which is described by figure 2. The chair configuration in figure 1 is most stable because there is no Di-axial interaction present. But in case of 2nd chair configuration there is 1,3-Di-axial interaction present.
Note:
1. The stability of chair configuration is found by adding up the A-value for each axial substituent. Lower the number, more stable it is.
2. The conformational isomers are defined as the stereoisomers which differ in relative position of atoms within the molecule and which can be interconverted simply by rotation about the sigma bond. In this the interconversion of the isomers does not require breaking and remaking of covalent bonds.
Complete step by step answer:
The structure of 1-tert-butyl-2- methylcyclohexane is given as below:

Now we will draw the two structure of chair conformation of 1-tert-butyl-2- methylcyclohexane as:

Figure 1.

Figure 2.
The most stable chair conformation structure of 1-tert-butyl-2- methylcyclohexane is that chair conformation which is described by figure 1. as compared to that chair configuration which is described by figure 2. The chair configuration in figure 1 is most stable because there is no Di-axial interaction present. But in case of 2nd chair configuration there is 1,3-Di-axial interaction present.
Note:
1. The stability of chair configuration is found by adding up the A-value for each axial substituent. Lower the number, more stable it is.
2. The conformational isomers are defined as the stereoisomers which differ in relative position of atoms within the molecule and which can be interconverted simply by rotation about the sigma bond. In this the interconversion of the isomers does not require breaking and remaking of covalent bonds.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




