Write the coordination number and oxidation state of platinum in the complex $\left[ {{{Pt}}{{\left( {{{en}}} \right)}_2}{{C}}{{{l}}_2}} \right]$.
Answer
610.8k+ views
Hint: Complex compounds are the compounds having complex ions. The given compound is an example of a complex compound. Coordination number defines the number of atoms or ligands which are directly bonded to the central metal ion or atom. The oxidation state defines the charge of the atom.
Complete step by step solution:
The structure of the given coordination compound is given below:

In the given compound, ${{en}}$ represents the compound ethylene diamine. Its chemical formula is ${{N}}{{{H}}_2}{{C}}{{{H}}_2}{{C}}{{{H}}_2}{{N}}{{{H}}_2}$. Its structure is given below:
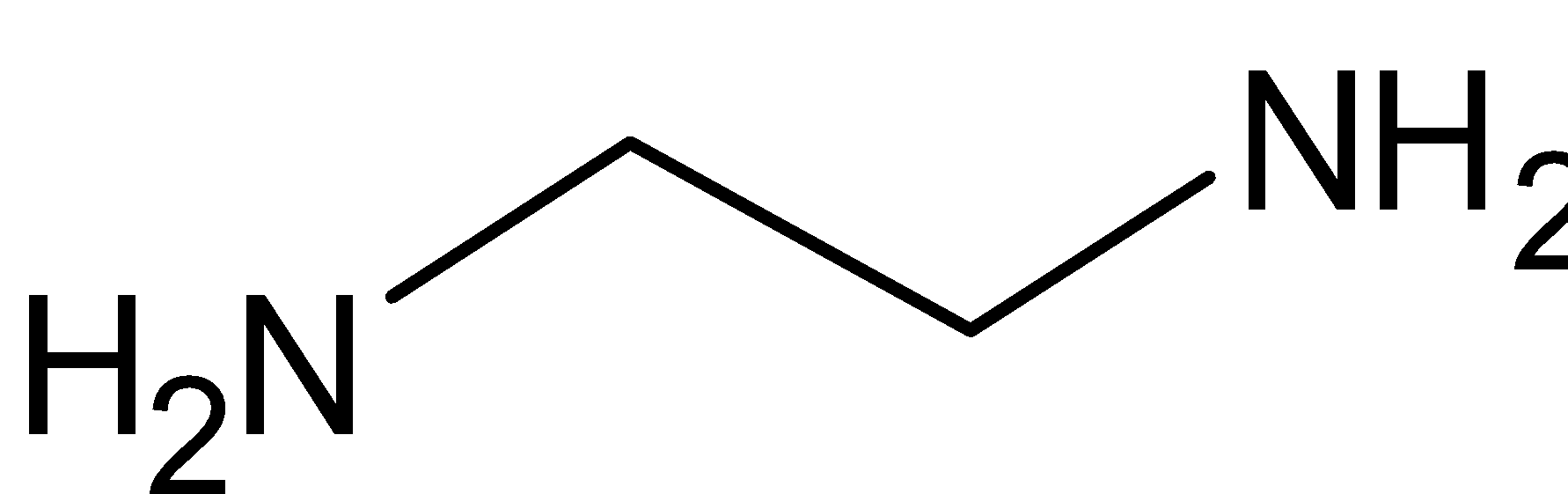
It is a bidentate ligand, i.e. it has two points of attachment or it can coordinate to metal at two points of the complex. Ethylene diamine has two nitrogen atoms which have two lone pairs of electrons. This is donated to the central metal ion. Also, the complex has two ethylene diamine ligands. Thus it donates a total of four electrons. And chlorine donates one electron each, so a total of two electrons from chlorine. Thus a total of six electrons are donated. So the coordination number of platinum is $6$.
Now let’s find the oxidation state of platinum. Oxidations state can be determined by knowing the charge on the complex ion and the ligand.
So let’s consider the oxidation state of platinum as ${{x}}$ and the charge on ethylene diamine is $0$ since it is a neutral ligand. And charge on chlorine ligands is $ - 1$. Combining all these values, we get
${{x}} + 0 + - 2 = 0 \Leftrightarrow {{x}} = + 2$.
Thus the oxidation number or oxidation state of platinum metal is $ + 2$ and its coordination number is $6$.
Note:
According to Werner’s theory, the primary valency is equivalent to the oxidation state while the secondary valency is equivalent to the coordination number. Primary valency can be varied. While secondary valency, for a certain oxidation, is fixed.
Complete step by step solution:
The structure of the given coordination compound is given below:

In the given compound, ${{en}}$ represents the compound ethylene diamine. Its chemical formula is ${{N}}{{{H}}_2}{{C}}{{{H}}_2}{{C}}{{{H}}_2}{{N}}{{{H}}_2}$. Its structure is given below:

It is a bidentate ligand, i.e. it has two points of attachment or it can coordinate to metal at two points of the complex. Ethylene diamine has two nitrogen atoms which have two lone pairs of electrons. This is donated to the central metal ion. Also, the complex has two ethylene diamine ligands. Thus it donates a total of four electrons. And chlorine donates one electron each, so a total of two electrons from chlorine. Thus a total of six electrons are donated. So the coordination number of platinum is $6$.
Now let’s find the oxidation state of platinum. Oxidations state can be determined by knowing the charge on the complex ion and the ligand.
So let’s consider the oxidation state of platinum as ${{x}}$ and the charge on ethylene diamine is $0$ since it is a neutral ligand. And charge on chlorine ligands is $ - 1$. Combining all these values, we get
${{x}} + 0 + - 2 = 0 \Leftrightarrow {{x}} = + 2$.
Thus the oxidation number or oxidation state of platinum metal is $ + 2$ and its coordination number is $6$.
Note:
According to Werner’s theory, the primary valency is equivalent to the oxidation state while the secondary valency is equivalent to the coordination number. Primary valency can be varied. While secondary valency, for a certain oxidation, is fixed.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




