With the reference of the scheme given, which of the given statement (s) about T, U, V and W is correct?
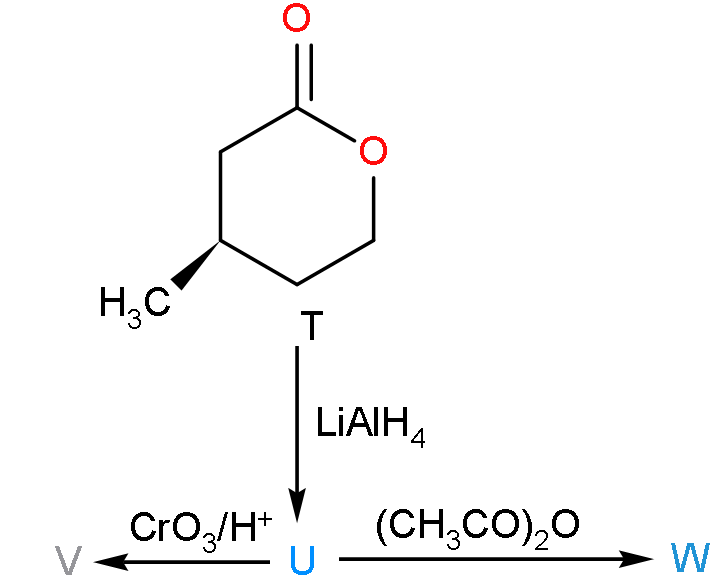
A. T is soluble in hot aqueous NaOH
B. U is optically active
C. Molecular formula of W is ${{\text{C}}_{10}}{{\text{H}}_{18}}{{\text{O}}_{4}}$
D. V gives effervescence on treatment with $\text{NaHC}{{\text{O}}_{3}}$

Answer
605.7k+ views
Hint: For this problem, firstly we have to write the product which will be formed when the compound T will react with Lithium aluminium hydroxide and then the further reaction to write the products V and W so that we can determine all the options.
Complete step by step answer:
- In the given question, we have to explain that among the given options which option is correct or not.
- Now, firstly the compound T will reduce when it will react with lithium aluminium hydroxide because it is a reducing agent so the product formed will be:
- Now, here the compound T when placed in the alkaline medium of sodium hydroxide then it will form a salt complex due to which it will be soluble.
- So, we can say that the compound T will be soluble in the hot aqueous solution of the sodium hydroxide.
- Now, when the compound U will react with acetic anhydride with highly acidic because it consists of two molecules of acetic acid.
- Then it will yield a compound as shown below:
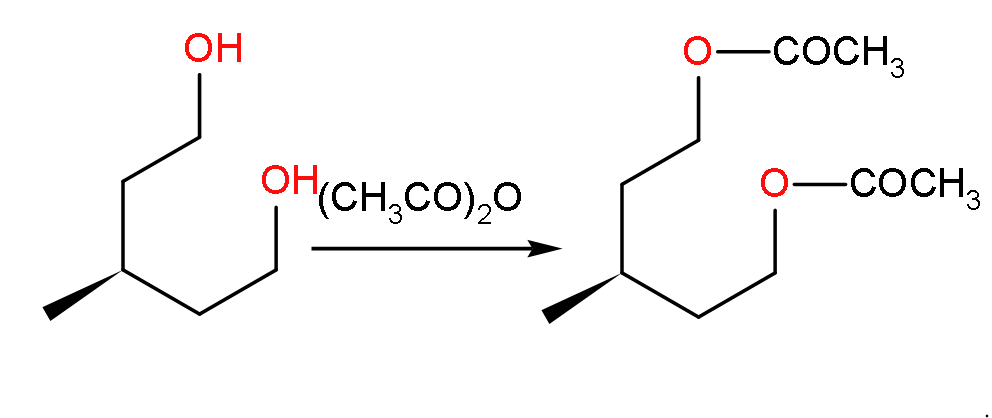
- So, here the product formed will have the molecular formula of ${{\text{C}}_{10}}{{\text{H}}_{18}}{{\text{O}}_{4}}$ because we can see in the structure that there are 10 carbon atoms, 18 hydrogen atoms and 4 oxygen atoms.
- So, we can say the statement C is correct.
- Now, firstly we have to find out the product V which is formed when the molecule U will undergo oxidation due to the presence of $\text{Cr}{{\text{O}}_{3}}/{{\text{H}}^{+}}$.
- So the product formed will be:
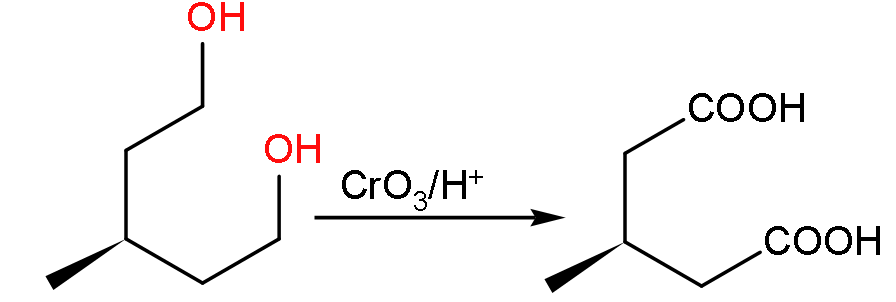
- Now, the molecular formula of the compound V will be ${{\text{C}}_{6}}{{\text{H}}_{10}}{{\text{O}}_{4}}$ so when it reacts with sodium carbonate it gives carbon dioxide gas.
- Due to the production of carbon dioxide gas it will show the effervescence as shown below:
${{\text{C}}_{6}}{{\text{H}}_{10}}{{\text{O}}_{4}}\text{ + NaHC}{{\text{O}}_{3}}\text{ }\to \text{ C}{{\text{O}}_{2}}\,\uparrow $
So, the correct answer is “Option A, C and D”.
Note: In this problem, the compound U is not the optically active compound because there is no chiral carbon present. As we know that the chiral carbon is the carbon which is attached to the four different groups.
Complete step by step answer:
- In the given question, we have to explain that among the given options which option is correct or not.
- Now, firstly the compound T will reduce when it will react with lithium aluminium hydroxide because it is a reducing agent so the product formed will be:
- Now, here the compound T when placed in the alkaline medium of sodium hydroxide then it will form a salt complex due to which it will be soluble.
- So, we can say that the compound T will be soluble in the hot aqueous solution of the sodium hydroxide.
- Now, when the compound U will react with acetic anhydride with highly acidic because it consists of two molecules of acetic acid.
- Then it will yield a compound as shown below:

- So, here the product formed will have the molecular formula of ${{\text{C}}_{10}}{{\text{H}}_{18}}{{\text{O}}_{4}}$ because we can see in the structure that there are 10 carbon atoms, 18 hydrogen atoms and 4 oxygen atoms.
- So, we can say the statement C is correct.
- Now, firstly we have to find out the product V which is formed when the molecule U will undergo oxidation due to the presence of $\text{Cr}{{\text{O}}_{3}}/{{\text{H}}^{+}}$.
- So the product formed will be:

- Now, the molecular formula of the compound V will be ${{\text{C}}_{6}}{{\text{H}}_{10}}{{\text{O}}_{4}}$ so when it reacts with sodium carbonate it gives carbon dioxide gas.
- Due to the production of carbon dioxide gas it will show the effervescence as shown below:
${{\text{C}}_{6}}{{\text{H}}_{10}}{{\text{O}}_{4}}\text{ + NaHC}{{\text{O}}_{3}}\text{ }\to \text{ C}{{\text{O}}_{2}}\,\uparrow $
So, the correct answer is “Option A, C and D”.
Note: In this problem, the compound U is not the optically active compound because there is no chiral carbon present. As we know that the chiral carbon is the carbon which is attached to the four different groups.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




