Which one of the following is strongest base in aqueous solution:
A. Trimethylamine
B. Aniline
C. Dimethylamine
D. Methylamine
Answer
609.9k+ views
Hint: The basicity is defined as the tendency of a molecule to donate the electrons or hydroxide ion. As the availability of the electrons increases, the basicity of the molecule increases.
Complete step by step answer:
In the amines, the nitrogen atom has a lone pair of electrons that can be donated, so the amines are basic.
Aliphatic amines are more basic than the aromatic amine.
Because in aromatic amine the phenyl ring works as an electron-withdrawing group which decreases the electron density on the nitrogen atom.
The $ - {\text{I}}$ effect in aniline is shown as follows:

In the case of aliphatic amines, the basicity is decided on the basis of steric hindrance and $ + {\text{I}}$ effect.
In the amine, the $- {\text{N}}{{\text{H}}_2}$ group is an electron-withdrawing group so, the $- {\text{N}}{{\text{H}}_2}$ group has $+ {\text{I}}$ effect. Thus, it withdraws the electron density from the alkyl chain so, as the number of alkyl groups increases, the basicity should be increased.
The $+ {\text{I}}$ effect in all aliphatic amines is shown as follows:
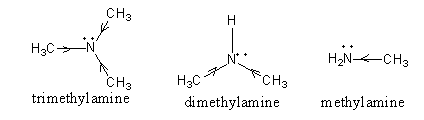
So, according to the $+ {\text{I}}$ effect, the basicity order should be,
${\text{trimethylamine}}\,{\text{ > }}\,{\text{dimethylamine}}\,{\text{ > }}\,{\text{methylamine}}$
As the steric hindrance in amine increases the hydrogen ion of water which takes an electron from amine cannot approach the amine so, the basicity decreases.
So, according to the steric hindrance, the basicity order should be,
$\,{\text{methylamine}}\,{\text{ > }}\,{\text{dimethylamine}}\,{\text{ > }}\,{\text{trimethylamine}}$
The order of basicity which comes by the combined effect of $ + {\text{I}}$ and steric hindrance is as follows:
$\,{\text{dimethylamine}}\,{\text{ > }}\,{\text{methylamine}}\,{\text{ > }}\,\,{\text{trimethylamine}}$
So, the maximum basicity is of dimethylamine.
Therefore, option (C) dimethylamine, is correct.
Note: Basicity is directly proportional to $+{\text{I}}$ effect and inversely proportional to the $-{\text{I}}$ and the steric hindrance. The basicity of aniline is minimum among all four due to $ - {\text{I}}$ of the ring.
Complete step by step answer:
In the amines, the nitrogen atom has a lone pair of electrons that can be donated, so the amines are basic.
Aliphatic amines are more basic than the aromatic amine.
Because in aromatic amine the phenyl ring works as an electron-withdrawing group which decreases the electron density on the nitrogen atom.
The $ - {\text{I}}$ effect in aniline is shown as follows:

In the case of aliphatic amines, the basicity is decided on the basis of steric hindrance and $ + {\text{I}}$ effect.
In the amine, the $- {\text{N}}{{\text{H}}_2}$ group is an electron-withdrawing group so, the $- {\text{N}}{{\text{H}}_2}$ group has $+ {\text{I}}$ effect. Thus, it withdraws the electron density from the alkyl chain so, as the number of alkyl groups increases, the basicity should be increased.
The $+ {\text{I}}$ effect in all aliphatic amines is shown as follows:

So, according to the $+ {\text{I}}$ effect, the basicity order should be,
${\text{trimethylamine}}\,{\text{ > }}\,{\text{dimethylamine}}\,{\text{ > }}\,{\text{methylamine}}$
As the steric hindrance in amine increases the hydrogen ion of water which takes an electron from amine cannot approach the amine so, the basicity decreases.
So, according to the steric hindrance, the basicity order should be,
$\,{\text{methylamine}}\,{\text{ > }}\,{\text{dimethylamine}}\,{\text{ > }}\,{\text{trimethylamine}}$
The order of basicity which comes by the combined effect of $ + {\text{I}}$ and steric hindrance is as follows:
$\,{\text{dimethylamine}}\,{\text{ > }}\,{\text{methylamine}}\,{\text{ > }}\,\,{\text{trimethylamine}}$
So, the maximum basicity is of dimethylamine.
Therefore, option (C) dimethylamine, is correct.
Note: Basicity is directly proportional to $+{\text{I}}$ effect and inversely proportional to the $-{\text{I}}$ and the steric hindrance. The basicity of aniline is minimum among all four due to $ - {\text{I}}$ of the ring.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




