Which one of the following halogen compounds is difficult to be hydrated by ${{S}_{N}}1$ mechanism?
(A) Tertiary butyl chloride
(B) Isopropyl chloride
(C) Benzyl Chloride
(D) Chlorobenzene
(E) Allyl chloride
Answer
602.4k+ views
Hint: For determining which of this halogen compound is difficult to be hydrated by ${{S}_{N}}1$ mechanism we need to find out in which of them halogen atom is firmly attached and cannot be replaced by nucleophile $O{{H}^ {-}} $ easily.
Complete step by step solution:
We have been provided with halogen compounds Tertiary butyl chloride, Isopropyl chloride, Benzyl Chloride, Chlorobenzene and Allyl chloride.
We need to find out which of the halogen compounds is difficult to be hydrated by ${{S}_{N}}1$ mechanism.
So, for that:
First, we have tertiary butyl chloride:
So, tertiary butyl chloride is unstable and can easily be hydrolyzed or hydrated${{S}_{N}}1$ mechanism.
Next, we have, isopropyl chloride:
Isopropyl chloride is unstable and can easily be hydrolyzed or hydrated by ${{S}_{N}}1$ mechanism.
Next, is benzyl chloride:
Benzyl chloride is unstable and can easily be hydrolyzed or hydrated by ${{S}_{N}}1$ mechanism.
Next, we have, chlorobenzene:
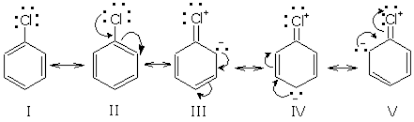
Chlorobenzene is quite stable due to the delocalization of electrons by resonance. Also, (C-Cl) bonds possess a double bond character due to which it is very difficult to hydrolyze by ${{S}_{N}}1$ mechanism.
Last one is Allyl chloride:
Allyl chloride is unstable and can easily be hydrolyzed or hydrated by ${{S}_{N}}1$ mechanism. So, we can say that chlorobenzene is very difficult to hydrolyze by ${{S}_{N}}1$ mechanism.
Therefore, we can conclude that option (D) is correct.
Note: The stability of resonance increases with the number of covalent bonds. Number of atoms with an octet of electrons. A negative charge if any on a more electronegative atom, a positive charge if any on the more electropositive atom, increases the stability of the atom.
Complete step by step solution:
We have been provided with halogen compounds Tertiary butyl chloride, Isopropyl chloride, Benzyl Chloride, Chlorobenzene and Allyl chloride.
We need to find out which of the halogen compounds is difficult to be hydrated by ${{S}_{N}}1$ mechanism.
So, for that:
First, we have tertiary butyl chloride:
So, tertiary butyl chloride is unstable and can easily be hydrolyzed or hydrated${{S}_{N}}1$ mechanism.
Next, we have, isopropyl chloride:
Isopropyl chloride is unstable and can easily be hydrolyzed or hydrated by ${{S}_{N}}1$ mechanism.
Next, is benzyl chloride:
Benzyl chloride is unstable and can easily be hydrolyzed or hydrated by ${{S}_{N}}1$ mechanism.
Next, we have, chlorobenzene:

Chlorobenzene is quite stable due to the delocalization of electrons by resonance. Also, (C-Cl) bonds possess a double bond character due to which it is very difficult to hydrolyze by ${{S}_{N}}1$ mechanism.
Last one is Allyl chloride:
Allyl chloride is unstable and can easily be hydrolyzed or hydrated by ${{S}_{N}}1$ mechanism. So, we can say that chlorobenzene is very difficult to hydrolyze by ${{S}_{N}}1$ mechanism.
Therefore, we can conclude that option (D) is correct.
Note: The stability of resonance increases with the number of covalent bonds. Number of atoms with an octet of electrons. A negative charge if any on a more electronegative atom, a positive charge if any on the more electropositive atom, increases the stability of the atom.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




