Which of the following substances is antiferromagnetic?
(a).$MnO$
(b).$Cr{{O}_{2}}$
(c).$F{{e}_{3}}{{O}_{4}}$
(d).$NaCl$
Answer
612.3k+ views
Hint: In materials that display antiferromagnetism, atom or molecular magnetic moments, typically associated with electron spins, align in a normal pattern with adjacent spins (on different sublattices) pointing in opposite directions.
Complete answer:
One component of the combined electromagnetic force is magnetism. It refers to physical effects induced by the force of magnets, objects creating fields that attract or repel other objects.
A magnetic domain is a region within a magnetic material where a uniform direction of magnetization is in. This implies the individual atomic magnetic moments are associated with each other and point in the same direction.
These are the products which are ferromagnetic, ferrimagnetic, and antiferromagnetic. There are no magnetic domains in paramagnetic and diamagnetic materials, in which the dipoles align in response to an external field but do not align spontaneously.
The$MnO$ has antiferromagnetism..
Anti-Ferromagnetism is a form of solid magnetism in which adjacent ions act as a tiny magnet. At low temperature, some ions align themselves.
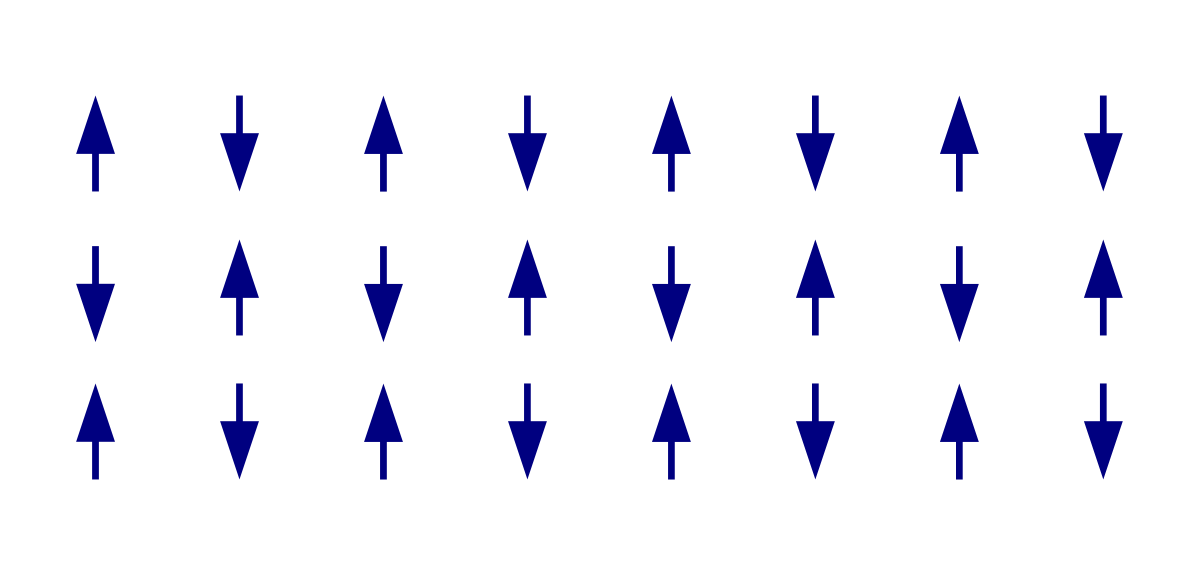
Adjacent ions that serve as small magnets (in this case manganese ions, $M{{n}^{2+}}$) bind themselves spontaneously to the opposite or antiparallel at relatively low temperatures.
$NaCl$ has a diamagnetic character. Diamagnetic materials are repelled by a magnetic field; an applied magnetic field creates in them in the opposite direction an induced magnetic field which causes repulsive force.
$Cr{{O}_{2}}$ has ferromagnetic properties. Ferromagnetism is the fundamental process by which certain materials shape, or are attracted to, permanent magnets.
$F{{e}_{3}}{{O}_{4}}$is ferrimagnetic at room temperature. Ferrimagnetic material is one that has populations of atoms with opposing magnetic moments, as in antiferromagnetism; however, the opposing moments are different in ferrimagnetic materials and there is a spontaneous magnetization.
Note:
-The root of magnetism lies in the electrons' orbital and spin motions, and how they interact with each other.
-Some may find that shocking, but all the matter is magnetic. It is just because some materials are much more magnetic than others. The principal difference is that there is no mutual interaction of atomic magnetic moments in some materials, while there is a very strong interaction between atomic moments in other materials.
Complete answer:
One component of the combined electromagnetic force is magnetism. It refers to physical effects induced by the force of magnets, objects creating fields that attract or repel other objects.
A magnetic domain is a region within a magnetic material where a uniform direction of magnetization is in. This implies the individual atomic magnetic moments are associated with each other and point in the same direction.
These are the products which are ferromagnetic, ferrimagnetic, and antiferromagnetic. There are no magnetic domains in paramagnetic and diamagnetic materials, in which the dipoles align in response to an external field but do not align spontaneously.
The$MnO$ has antiferromagnetism..
Anti-Ferromagnetism is a form of solid magnetism in which adjacent ions act as a tiny magnet. At low temperature, some ions align themselves.

Adjacent ions that serve as small magnets (in this case manganese ions, $M{{n}^{2+}}$) bind themselves spontaneously to the opposite or antiparallel at relatively low temperatures.
$NaCl$ has a diamagnetic character. Diamagnetic materials are repelled by a magnetic field; an applied magnetic field creates in them in the opposite direction an induced magnetic field which causes repulsive force.
$Cr{{O}_{2}}$ has ferromagnetic properties. Ferromagnetism is the fundamental process by which certain materials shape, or are attracted to, permanent magnets.
$F{{e}_{3}}{{O}_{4}}$is ferrimagnetic at room temperature. Ferrimagnetic material is one that has populations of atoms with opposing magnetic moments, as in antiferromagnetism; however, the opposing moments are different in ferrimagnetic materials and there is a spontaneous magnetization.
Note:
-The root of magnetism lies in the electrons' orbital and spin motions, and how they interact with each other.
-Some may find that shocking, but all the matter is magnetic. It is just because some materials are much more magnetic than others. The principal difference is that there is no mutual interaction of atomic magnetic moments in some materials, while there is a very strong interaction between atomic moments in other materials.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

The correct structure of ethylenediaminetetraacetic class 12 chemistry CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

How many atoms of XeO64 lie in the same plane class 12 chemistry CBSE

Maltose is a reducing sugar whereas sucrose is a nonreducing class 12 biology CBSE

How many states of matter are there in total class 12 chemistry CBSE




