Which of the following statements is/are not false? (This question has multiple correct answers).
(a)- Sorbitol and mannitol are epimers
(b)- Sorbitol and mannitol do not show the relationship as an object and its mirror image
(c)- Sorbitol and mannitol differ in the number of –OH group in the molecule
(d)- All of the above
Answer
537.6k+ views
Hint: Both sorbitol and mannitol are carbohydrates in which there are six carbon atoms. epimers are those compounds if they differ in configuration only at one carbon atom.
Complete answer:
Sorbitol and Mannitol are both carbohydrates because they are made up of carbon, hydrogen, and oxygen atoms. The formula of both sorbitol and mannitol are the same, and its formula is ${{C}_{6}}{{H}_{14}}{{O}_{6}}$.
Since the formula of sorbitol and mannitol are the same but the arrangements of the groups around the carbon atoms are different. The structures of them are given below:
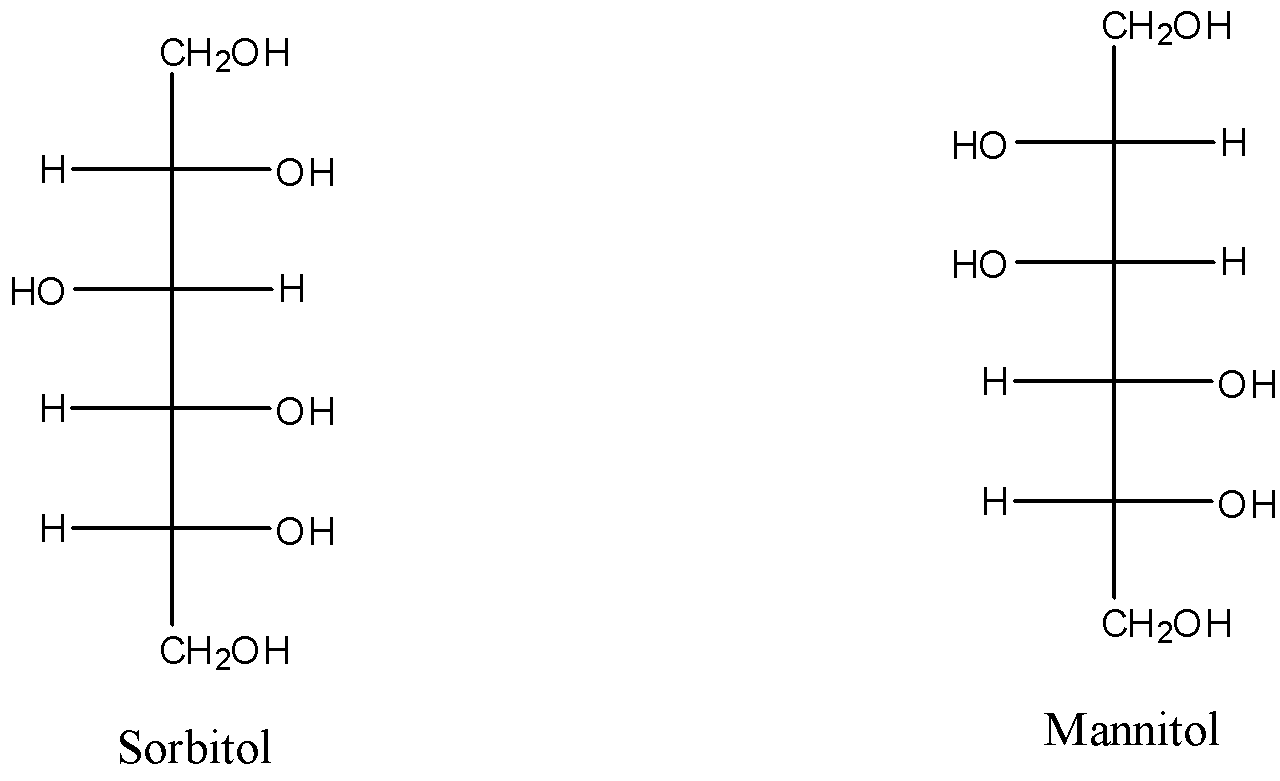
As we can see that there is only one change in the configuration at one carbon atom. So, they are epimers. Therefore, statement (a) is correct.
The relationship between an item and its reflection, since the configuration is uniform to one single carbon atom, cannot be shown in Sorbitol and mannitol. So, statement (b) is also correct.
Both sorbitol and mannitol have six hydroxyl groups (-OH), only the arrangements of the groups are different. So, statement (c) is incorrect.
Therefore, from the given options, option (a) and option (b) are correct answers.
Note:
Both sorbitol and mannitol are sugar alcohol and are isomers of each other. Their properties like uses, melting points, and sources are different even though they look similar.
Complete answer:
Sorbitol and Mannitol are both carbohydrates because they are made up of carbon, hydrogen, and oxygen atoms. The formula of both sorbitol and mannitol are the same, and its formula is ${{C}_{6}}{{H}_{14}}{{O}_{6}}$.
Since the formula of sorbitol and mannitol are the same but the arrangements of the groups around the carbon atoms are different. The structures of them are given below:

As we can see that there is only one change in the configuration at one carbon atom. So, they are epimers. Therefore, statement (a) is correct.
The relationship between an item and its reflection, since the configuration is uniform to one single carbon atom, cannot be shown in Sorbitol and mannitol. So, statement (b) is also correct.
Both sorbitol and mannitol have six hydroxyl groups (-OH), only the arrangements of the groups are different. So, statement (c) is incorrect.
Therefore, from the given options, option (a) and option (b) are correct answers.
Note:
Both sorbitol and mannitol are sugar alcohol and are isomers of each other. Their properties like uses, melting points, and sources are different even though they look similar.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Name the part of the flower which the tassels of the class 12 biology CBSE

The value of constant k in coulombs law is beginalign class 12 physics CBSE

Suicide bags of cells are aEndoplasmic reticulum bLysosome class 12 biology CBSE

Quantisation of charge implies a Charge does not exist class 12 physics CBSE




