Which of the following oxides of nitrogen is a coloured gas?
A. ${N_2}O$
B.NO
C. ${N_2}{O_4}$
D. $N{O_2}$
Answer
609k+ views
Hint: Nitrogen dioxide appears as a reddish-brown gas. $N{O_2}$ along with aerosols, is responsible for the reddish - brown colour of smog. Nitrogen dioxide partners to solid dinitrogen tetroxide at low temperatures.
Complete step by step answer:
Nitrous Oxide $({N_2}O)$ or dinitrogen oxide is also known as Laughing Gas. Nitrous Oxide is a naturally occurring gas that is colourless and sweet-tasting gas. It is a non-flammable gas.

Nitric Oxide (NO) is a colourless gas. It achieves blue shade when it is melted. It is sparingly solvent in water and flammable gas with a slight odour. It is a molecule with a linear shape and is a resonance hybrid of the two structures. The bond length of NO is 115 pm which is intermediate between double bond and triple bond. And it is also paramagnetic in nature.
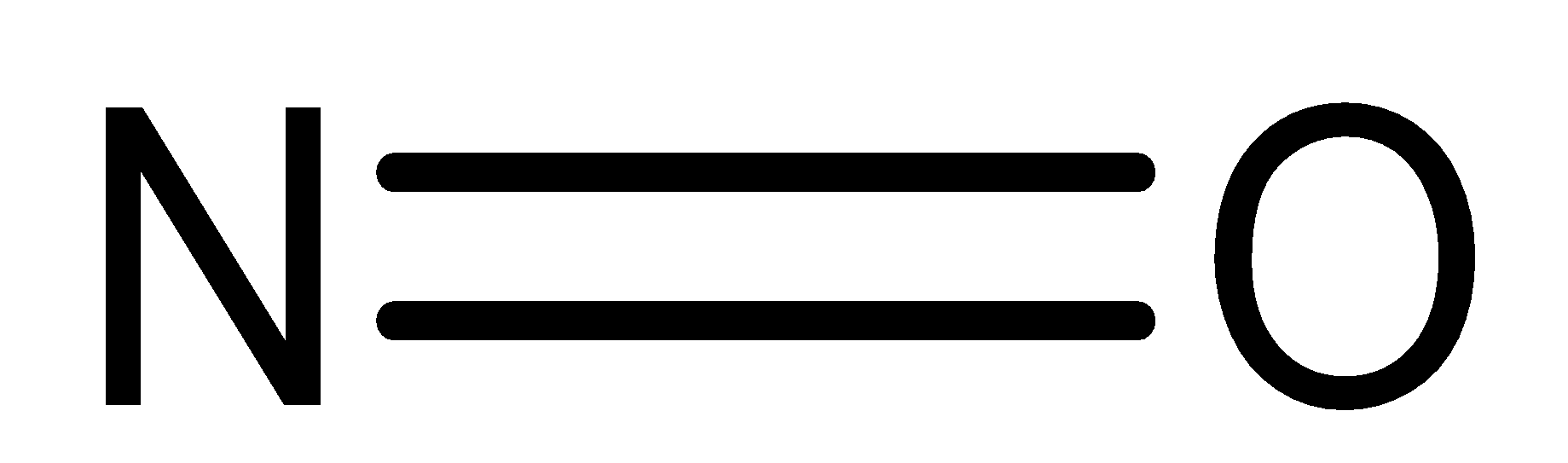
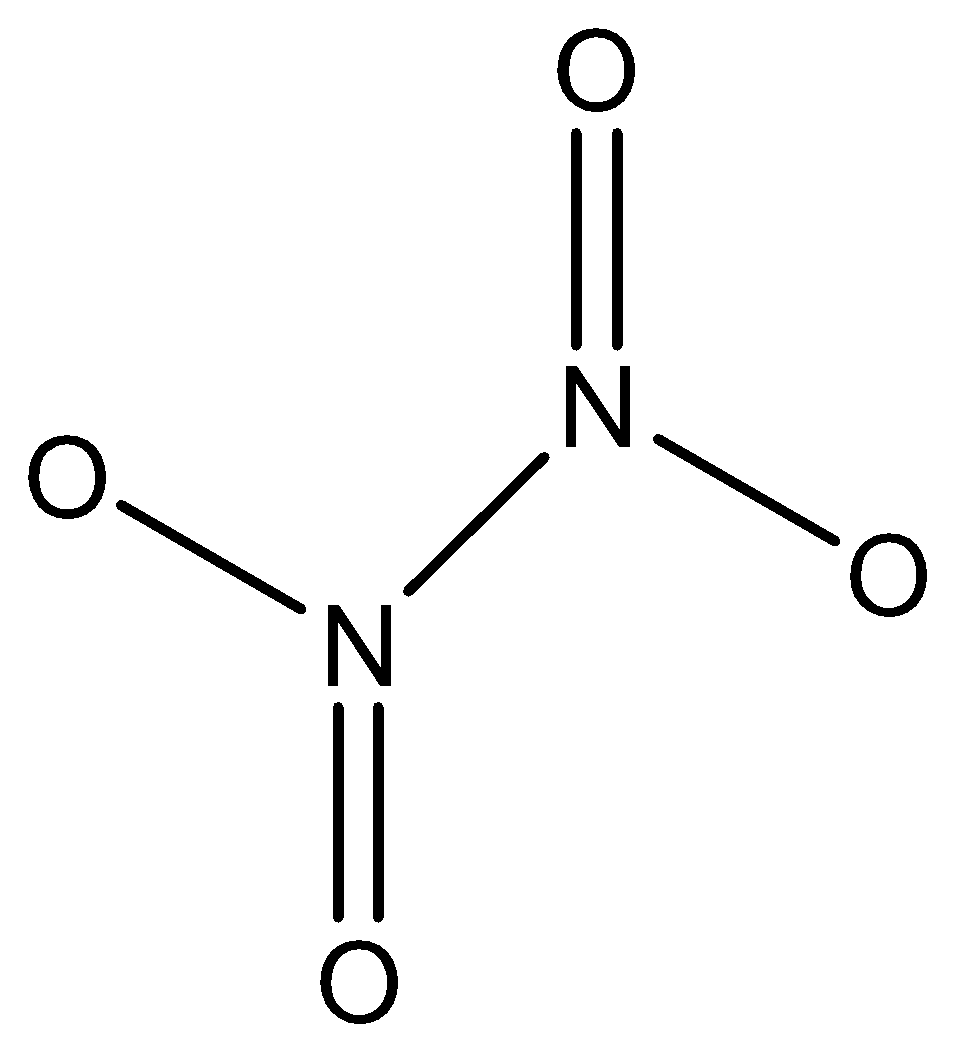
Dinitrogen tetroxide $({N_2}{O_4})$ is a colourless gas. It has an irritating, unpleasant acid-like odour. ${N_2}{O_4}$ is a very reactive, toxic oxidizer. It is non-flammable with air but it will inflame combustible materials.
Nitrogen dioxide is a dark reddish-brown gas. It is poisonous but not flammable. $N{O_2}$ along with aerosols, is responsible for the reddish - brown colour of smog. At high concentrations it is highly toxic, and can cause serious lung damage. Nitrogen dioxide is also a strong oxidising agent and thus it is very reactive with other compounds.

Therefore, the correct answer is option (D).
Note: Nitrogen dioxide is an acidic oxide with angular shape. Its bond angle is 134 degrees and bond length is 120 pm. The atom is found as a resonance hybrid and is a paramagnetic substance. Because of the nearness of unpaired electrons, it dimerizes to a colourless dinitrogen tetroxide $({N_2}{O_4})$ atom that has an even number of electrons.
Complete step by step answer:
Nitrous Oxide $({N_2}O)$ or dinitrogen oxide is also known as Laughing Gas. Nitrous Oxide is a naturally occurring gas that is colourless and sweet-tasting gas. It is a non-flammable gas.

Nitric Oxide (NO) is a colourless gas. It achieves blue shade when it is melted. It is sparingly solvent in water and flammable gas with a slight odour. It is a molecule with a linear shape and is a resonance hybrid of the two structures. The bond length of NO is 115 pm which is intermediate between double bond and triple bond. And it is also paramagnetic in nature.
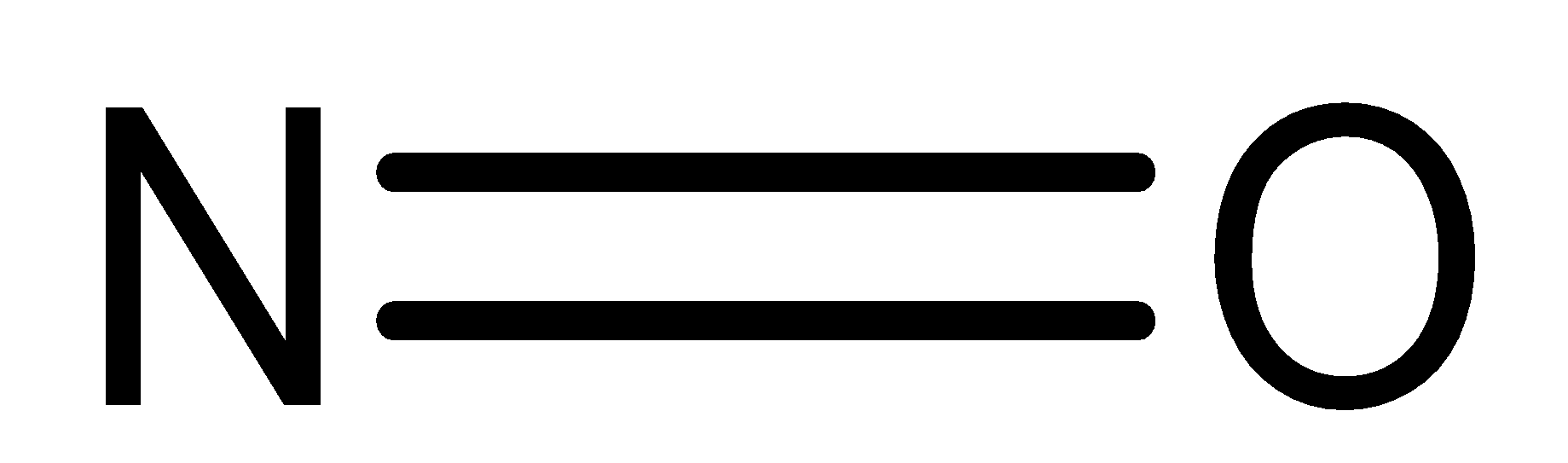
Dinitrogen tetroxide $({N_2}{O_4})$ is a colourless gas. It has an irritating, unpleasant acid-like odour. ${N_2}{O_4}$ is a very reactive, toxic oxidizer. It is non-flammable with air but it will inflame combustible materials.

Nitrogen dioxide is a dark reddish-brown gas. It is poisonous but not flammable. $N{O_2}$ along with aerosols, is responsible for the reddish - brown colour of smog. At high concentrations it is highly toxic, and can cause serious lung damage. Nitrogen dioxide is also a strong oxidising agent and thus it is very reactive with other compounds.

Therefore, the correct answer is option (D).
Note: Nitrogen dioxide is an acidic oxide with angular shape. Its bond angle is 134 degrees and bond length is 120 pm. The atom is found as a resonance hybrid and is a paramagnetic substance. Because of the nearness of unpaired electrons, it dimerizes to a colourless dinitrogen tetroxide $({N_2}{O_4})$ atom that has an even number of electrons.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




