Which of the following is not true about amino acids?
(a)- They are constituents of all proteins.
(b)- Alanine having one amino and one carboxyl group.
(c)- Most naturally occurring amino acids have D-configuration.
(d)- Glycine is the only naturally occurring amino acid which is optically inactive.
Answer
624.6k+ views
Hint: α-amino acids are the building blocks of protein. The optical activity of the compound is checked by checking whether the compound has a chiral carbon atom. The amino acid is classified into neutral, acidic, and basic.
Complete answer:
Let us study all the options one by one:
(a)- They are constituents of all proteins.
Chemically proteins are condensation polymers (actual polyamides) in which the monomeric units are the \[\alpha -\text{amino acids}\]. All the proteins contain the elements carbon, nitrogen, oxygen, sulfur, iron, copper, etc, on partial hydrolysis gives peptides and on complete hydrolysis gives \[\alpha -\text{amino acids}\].
(b)- Alanine having one amino and one carboxyl group.
\[\alpha -\text{amino acids}\]can be classified into three classes- neutral, acidic, and basic amino acids. Neutral are those which have one amino and one carboxyl group. Alanine comes under this group. Acid is those which have more carboxyl groups and basic are those which have more amino groups.
(c)- Most naturally occurring amino acids have D-configuration.
All the naturally occurring amino acids belong to the L-series. In other words, a protein exclusively consists of L-amino acids. In contrast, all the sugar belongs to D-series.
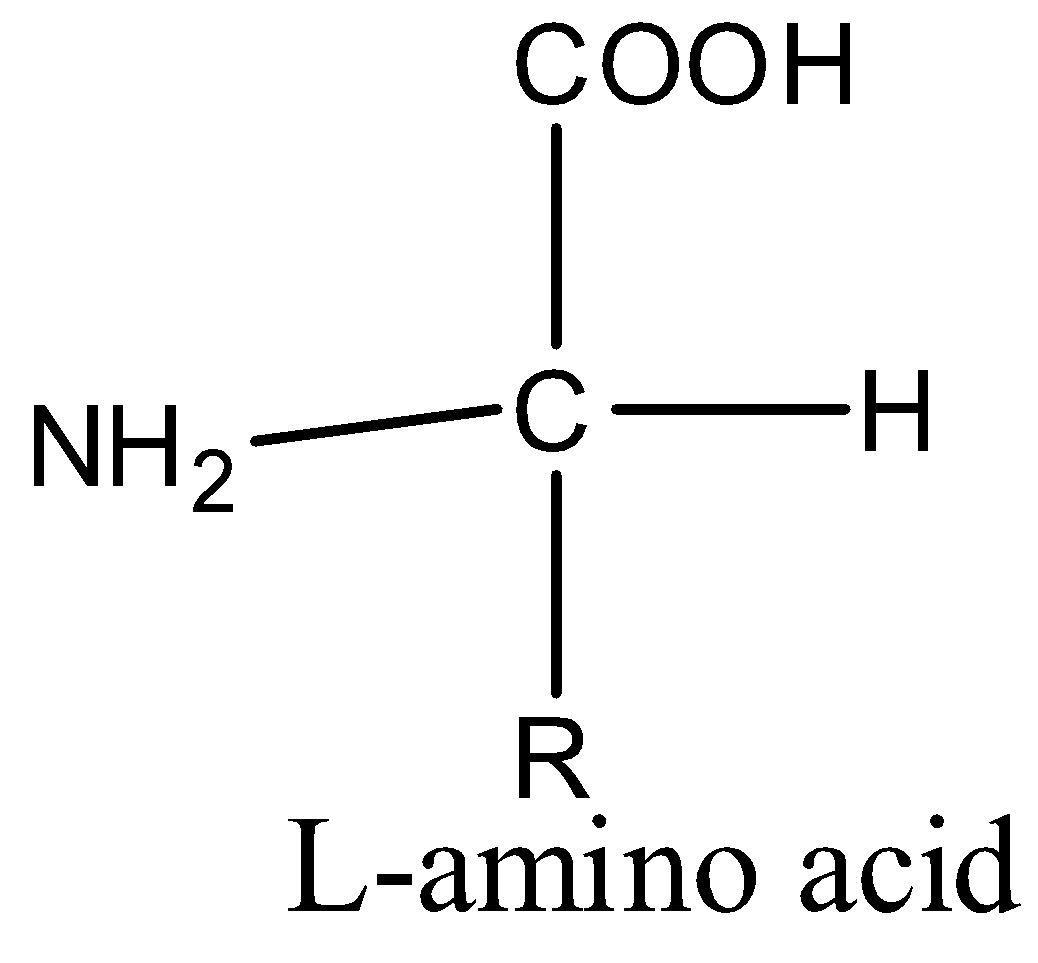
(d)- Glycine is the only naturally occurring amino acid which is optically inactive.
All the amino acids are optically active because they have chiral carbon atoms i.e., a carbon atom which has all the four substituents different. But in glycine, the carbon atom has one amino, one carboxyl, and two hydrogen groups.
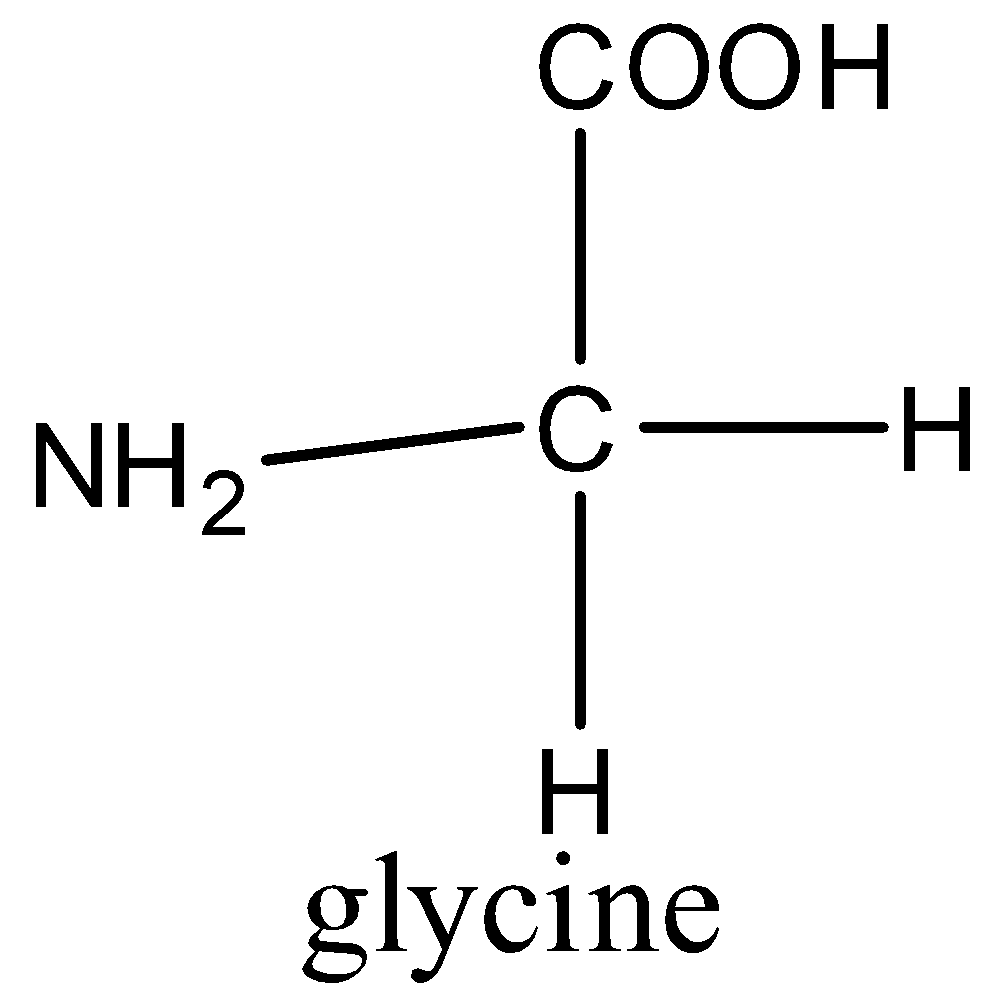
So, the correct answer is “Option C”.
Note: There are some exceptions of amino acids that must be kept in mind while reasoning these questions. For checking the optical activity the chiral carbon should be checked, if only one chiral is present in the compound then it is optically active.
Complete answer:
Let us study all the options one by one:
(a)- They are constituents of all proteins.
Chemically proteins are condensation polymers (actual polyamides) in which the monomeric units are the \[\alpha -\text{amino acids}\]. All the proteins contain the elements carbon, nitrogen, oxygen, sulfur, iron, copper, etc, on partial hydrolysis gives peptides and on complete hydrolysis gives \[\alpha -\text{amino acids}\].
(b)- Alanine having one amino and one carboxyl group.
\[\alpha -\text{amino acids}\]can be classified into three classes- neutral, acidic, and basic amino acids. Neutral are those which have one amino and one carboxyl group. Alanine comes under this group. Acid is those which have more carboxyl groups and basic are those which have more amino groups.
(c)- Most naturally occurring amino acids have D-configuration.
All the naturally occurring amino acids belong to the L-series. In other words, a protein exclusively consists of L-amino acids. In contrast, all the sugar belongs to D-series.

(d)- Glycine is the only naturally occurring amino acid which is optically inactive.
All the amino acids are optically active because they have chiral carbon atoms i.e., a carbon atom which has all the four substituents different. But in glycine, the carbon atom has one amino, one carboxyl, and two hydrogen groups.

So, the correct answer is “Option C”.
Note: There are some exceptions of amino acids that must be kept in mind while reasoning these questions. For checking the optical activity the chiral carbon should be checked, if only one chiral is present in the compound then it is optically active.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

A dentist uses a small mirror that gives a magnification class 12 physics CBSE




