Which of the following is not D sugar?
A)
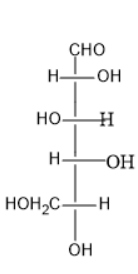
B)
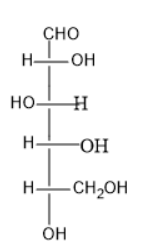
C)
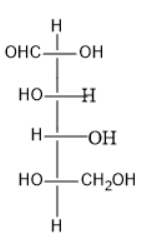
D)
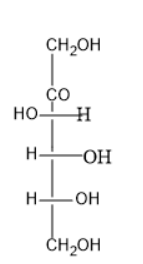




Answer
523.5k+ views
Hint: Carbohydrates are sugar molecules that have asymmetric carbons and are classified into D and L configurations.
D configuration can be given to sugars containing hydroxyl groups right to the farthest asymmetric carbon from the carbonyl group.
L configuration can be given to sugars containing hydroxyl groups left to the farthest asymmetric carbon from the carbonyl group.
Complete answer: Carbohydrates are sugar molecules consisting of mainly carbon, hydrogen and oxygen atoms.
These are also called hydrates of carbon.
The general empirical formula of carbohydrates is \[{C_m}{\left( {{H_2}O} \right)_n}\]
The oxygen and hydrogen are in the ratio of 1:2 in the carbohydrates.
Carbohydrates contain carboxyl and hydroxyl groups.
Based on the hydroxyl group, these sugars are classified into D and L sugars.
The sugar molecule with the asymmetric carbon farther from the carbonyl group contains the hydroxyl group at the left side; it can be called L sugar.
The sugar molecule with the asymmetric carbon farther from the carbonyl group contains the hydroxyl group at the right side; it can be called L sugar.
In the given options,
Option (A) contains five carbon atoms; the last carbon is farther from the carbonyl group than the hydroxyl group at the left side.
Thus, it is L sugar.
Option (B) contains five carbon atoms; the last carbon is farther from the carbonyl group than the hydroxyl group at right side.
Thus, it is D sugar.
Option (C) contains five carbon atoms; the last carbon is farther from the carbonyl group and contains the hydroxyl group at both right and left side.
Thus, it is neither L nor D sugar.
Option (D) contains five carbon atoms; the last carbon is farther from the carbonyl group than the hydroxyl group at right side.
Thus, it is D sugar.
Thus, option A is L-sugar.
Note:
The configuration must be written by considering the farthest carbon from the carbonyl group.
Though the nearest carbons contain hydroxyl groups, they should not be considered.
The direction of the hydroxyl group to the last farther carbon from the carbonyl group must be clearly noticed.
D configuration can be given to sugars containing hydroxyl groups right to the farthest asymmetric carbon from the carbonyl group.
L configuration can be given to sugars containing hydroxyl groups left to the farthest asymmetric carbon from the carbonyl group.
Complete answer: Carbohydrates are sugar molecules consisting of mainly carbon, hydrogen and oxygen atoms.
These are also called hydrates of carbon.
The general empirical formula of carbohydrates is \[{C_m}{\left( {{H_2}O} \right)_n}\]
The oxygen and hydrogen are in the ratio of 1:2 in the carbohydrates.
Carbohydrates contain carboxyl and hydroxyl groups.
Based on the hydroxyl group, these sugars are classified into D and L sugars.
The sugar molecule with the asymmetric carbon farther from the carbonyl group contains the hydroxyl group at the left side; it can be called L sugar.
The sugar molecule with the asymmetric carbon farther from the carbonyl group contains the hydroxyl group at the right side; it can be called L sugar.
In the given options,
Option (A) contains five carbon atoms; the last carbon is farther from the carbonyl group than the hydroxyl group at the left side.
Thus, it is L sugar.
Option (B) contains five carbon atoms; the last carbon is farther from the carbonyl group than the hydroxyl group at right side.
Thus, it is D sugar.
Option (C) contains five carbon atoms; the last carbon is farther from the carbonyl group and contains the hydroxyl group at both right and left side.
Thus, it is neither L nor D sugar.
Option (D) contains five carbon atoms; the last carbon is farther from the carbonyl group than the hydroxyl group at right side.
Thus, it is D sugar.
Thus, option A is L-sugar.
Note:
The configuration must be written by considering the farthest carbon from the carbonyl group.
Though the nearest carbons contain hydroxyl groups, they should not be considered.
The direction of the hydroxyl group to the last farther carbon from the carbonyl group must be clearly noticed.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




