Which of the following is not an aromatic compound?
A)

B)
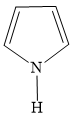
C)
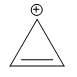
D)
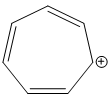




Answer
602.7k+ views
Hint: Follow the Huckel rule of aromaticity. A conjugated, cyclic planar compound having \[\left( {4n + 2} \right){\text{ }}\pi \] electrons is aromatic.
Complete answer:
If the number of pi electrons in the compound is not equal to \[\left( {4n + 2} \right)\] then the compound is non aromatic. For \[n = 0,1,2,3...\] the value of \[\left( {4n + 2} \right)\] is \[4\left( 0 \right) + 2 = 2,4\left( 1 \right) + 2 = 6,{\text{ }}4\left( 2 \right) + 2 = 10,{\text{ }}4\left( 3 \right) + 2 = 14...\] respectively.
Hence, if the number of pi electrons in a compound is not equal to 2,6,10, 14… then it is not an aromatic compound. In the option A ) cycloheptatrienyl anion has 8 pi electrons. Thus the number of pi electrons in cycloheptatrienyl anion is not equal to 2,6,10, 14…
Hence, cycloheptatrienyl anion is not an aromatic compound.
Hence, the correct option is option A ).
Additional Information: The compounds/ions in options B ) and D ) have 6 pi electrons. They are cyclic, planar and conjugated systems of pi electrons. Hence, compounds/ions of options B ) and D ) are aromatic.
The ion of option C ) has 2 pi electrons. It is conjugated, cyclic and planar. Hence, it is aromatic in nature.
Note: While counting the number of pi electrons, each double bond corresponds to a pair of electrons. Each lone pair of electrons corresponds to a pair of electrons. The orbital in which the lone pair of electrons is present, should be parallel to the p orbitals containing pi electrons. If a molecule/ion contains \[\left( {4n + 2} \right){\text{ }}\pi \] pi electrons, but it is non cyclic, non planar or non conjugated, then it is not an aromatic compound.
Complete answer:
If the number of pi electrons in the compound is not equal to \[\left( {4n + 2} \right)\] then the compound is non aromatic. For \[n = 0,1,2,3...\] the value of \[\left( {4n + 2} \right)\] is \[4\left( 0 \right) + 2 = 2,4\left( 1 \right) + 2 = 6,{\text{ }}4\left( 2 \right) + 2 = 10,{\text{ }}4\left( 3 \right) + 2 = 14...\] respectively.
Hence, if the number of pi electrons in a compound is not equal to 2,6,10, 14… then it is not an aromatic compound. In the option A ) cycloheptatrienyl anion has 8 pi electrons. Thus the number of pi electrons in cycloheptatrienyl anion is not equal to 2,6,10, 14…
Hence, cycloheptatrienyl anion is not an aromatic compound.
Hence, the correct option is option A ).
Additional Information: The compounds/ions in options B ) and D ) have 6 pi electrons. They are cyclic, planar and conjugated systems of pi electrons. Hence, compounds/ions of options B ) and D ) are aromatic.
The ion of option C ) has 2 pi electrons. It is conjugated, cyclic and planar. Hence, it is aromatic in nature.
Note: While counting the number of pi electrons, each double bond corresponds to a pair of electrons. Each lone pair of electrons corresponds to a pair of electrons. The orbital in which the lone pair of electrons is present, should be parallel to the p orbitals containing pi electrons. If a molecule/ion contains \[\left( {4n + 2} \right){\text{ }}\pi \] pi electrons, but it is non cyclic, non planar or non conjugated, then it is not an aromatic compound.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

Which among the following are examples of coming together class 11 social science CBSE




