Which of the following is not a nucleophile?
A. $C{{H}_{3}}{{O}^{-}}$
B. ${{H}_{2}}O$
C. $C{{H}_{3}}-OC{{H}_{3}}$
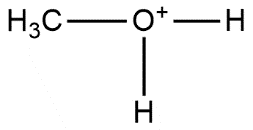
D.

Answer
568.8k+ views
Hint: A nucleophile is generally made up of two words Nucleo and phile where Nucleo refers to nucleus and phile is a Greek word having the meaning love so nucleophiles are also termed as those substances which are nucleus loving.
Complete step by step answer:
- Nucleophiles are generally said to be electron-rich species i.e. those species which have a greater tendency to donate electrons. Due to the nature of donating electrons nucleophiles are kept in the category of Lewis bases as bases are those substances that can donate the \[{{H}^{+}}\] ions. Nucleophiles either carry a negative charge or are neutral in nature.
- The nucleophilic nature of any species describes the affinity of the species towards the positively charged nucleus. All molecules or ions which have a free pair of electrons or at least one pi bond can act as a nucleophile.
- Hence from the above discussion, we can consider that nucleophiles are a chemical species which have the tendency to donate electron pairs to form a chemical bond in a reaction, it can be negative or neutral in charge. From this definition, we can easily conclude that option D is not suitable with a definition of the nucleophile. So, the correct answer is “Option D”.
Note: Nucleophiles generally take part in the nucleophilic substitution reactions and during this reaction nucleophile becomes attracted towards a partial or full positive charge rather than this neutral nucleophilic reaction with solvents like water is known by the name solvolysis.
Complete step by step answer:
- Nucleophiles are generally said to be electron-rich species i.e. those species which have a greater tendency to donate electrons. Due to the nature of donating electrons nucleophiles are kept in the category of Lewis bases as bases are those substances that can donate the \[{{H}^{+}}\] ions. Nucleophiles either carry a negative charge or are neutral in nature.
- The nucleophilic nature of any species describes the affinity of the species towards the positively charged nucleus. All molecules or ions which have a free pair of electrons or at least one pi bond can act as a nucleophile.
- Hence from the above discussion, we can consider that nucleophiles are a chemical species which have the tendency to donate electron pairs to form a chemical bond in a reaction, it can be negative or neutral in charge. From this definition, we can easily conclude that option D is not suitable with a definition of the nucleophile. So, the correct answer is “Option D”.
Note: Nucleophiles generally take part in the nucleophilic substitution reactions and during this reaction nucleophile becomes attracted towards a partial or full positive charge rather than this neutral nucleophilic reaction with solvents like water is known by the name solvolysis.
Recently Updated Pages
Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Explain the structure of megasporangium class 12 biology CBSE

What are the major means of transport Explain each class 12 social science CBSE

How many chromosomes are found in human ovum a 46 b class 12 biology CBSE

The diagram of the section of a maize grain is given class 12 biology CBSE




