Which of the following is an example of condensation polymer?
(A) Polythene
(B) Orlon
(C) Terylene
(D) None of these
Answer
612.9k+ views
Hint: Think about the formation of condensation polymers. Look carefully at the names of the three polymers given in the options and then try writing their synthesis. Identify the polymer which is prepared from condensation polymerization reactions.
Complete step by step answer:
- The process of synthesis of polymers is known as polymerization.
- Condensation polymerization is the process in which polymers are formed by condensation of two or more monomers by elimination of simple molecules like water or methanol. The monomers used have bifunctional groups. In these reactions, the product formed in the first step has two functional groups to continue and increase the chain. It is also called Step growth polymerization.
- A polymer obtained by condensation polymerization is known as Condensation polymer.
- Terylene is a condensation polymer. Terylene is synthesized using ethylene glycol and dimethyl terephthalate in the presence of zinc acetate and antimony trioxide as catalysts at 503K. Transesterification takes place and then polymerization to give terylene.
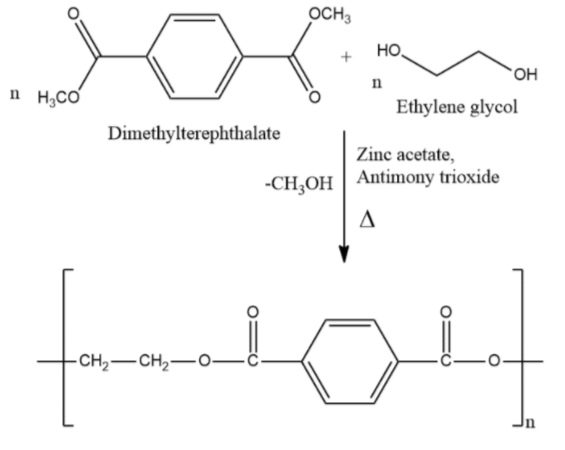
- Polythene and Orlon are addition polymers formed by chain growth polymerization.
- Polythene as the name suggests has is made up of only one monomer that is ethane.
- Orlon is also called acrilan. It has acrylonitrile as monomeric units.
- Both polythene and orlon are formed from free radical polymerization.
- Therefore, terylene is an example of condensation polymer.
So, the correct answer is “Option C”.
Note: Terylene is a step growth polymer. Terylene is also known as dacron or polyester. Terylene fibers are resistant to heat, crease, chemicals, light, moths, bacteria, etc. They absorb very less water and dry quickly. They are blended to cotton to form terrycot and with wool to form terry wool. Terylene is also used in preparation of fishing nets, ropes, trousers, magnetic tape recorders, tyres, etc.
Complete step by step answer:
- The process of synthesis of polymers is known as polymerization.
- Condensation polymerization is the process in which polymers are formed by condensation of two or more monomers by elimination of simple molecules like water or methanol. The monomers used have bifunctional groups. In these reactions, the product formed in the first step has two functional groups to continue and increase the chain. It is also called Step growth polymerization.
- A polymer obtained by condensation polymerization is known as Condensation polymer.
- Terylene is a condensation polymer. Terylene is synthesized using ethylene glycol and dimethyl terephthalate in the presence of zinc acetate and antimony trioxide as catalysts at 503K. Transesterification takes place and then polymerization to give terylene.

- Polythene and Orlon are addition polymers formed by chain growth polymerization.
- Polythene as the name suggests has is made up of only one monomer that is ethane.
- Orlon is also called acrilan. It has acrylonitrile as monomeric units.
- Both polythene and orlon are formed from free radical polymerization.
- Therefore, terylene is an example of condensation polymer.
So, the correct answer is “Option C”.
Note: Terylene is a step growth polymer. Terylene is also known as dacron or polyester. Terylene fibers are resistant to heat, crease, chemicals, light, moths, bacteria, etc. They absorb very less water and dry quickly. They are blended to cotton to form terrycot and with wool to form terry wool. Terylene is also used in preparation of fishing nets, ropes, trousers, magnetic tape recorders, tyres, etc.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




