Which of the following is an antiaromatic compound?
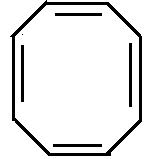
A.
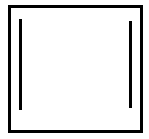
B.
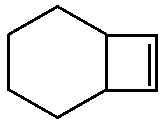
C.
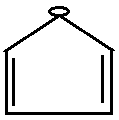
D.




Answer
564k+ views
Hint: We need to find the anti- aromatic compound. For solving this we use the aromaticity rule. First, the compound to be observed must be cyclic. Also, every atom in the ring must be conjugated. Then the molecule must have $ \left( {4n + 2} \right)\pi $ electrons. Anti-aromatic compounds have 4n pi electrons.
Complete step by step answer:
We will start by mentioning the aromaticity rule.
This rule is followed by aromatic and anti- aromatic compounds.
First, the compound to be observed must be cyclic. Also, every atom in the ring must be conjugated. Then the molecule must have $ \left( {4n + 2} \right)\pi $ electrons. At last, the molecule must be flat, but there are rare exceptions.
Option A is cyclooctatriene which is a non-aromatic compound. It changes to tub shaped structure.
Option B is cyclobutadiene which has $ {\text{2 \times n}}{{\text{e}}^{\text{ - }}}{\text{ where n = 2 = 4}}{{\text{e}}^{\text{ - }}} $ ,so it is antiaromatic.
Option C is not planar as the two middle C atoms are $ s{p^3} $ hybridised, thus making it non planar.
Option D is pentagon with negative charge and it is aromatic as it has $ {\text{2n}}{{\text{e}}^{\text{ - }}}{\text{ where n = 3}} $ .
Now we need to select the correct option.
Thus, the correct option is B.
Note:
Aromatic compounds are chemical compounds that consist of conjugated planar ring systems accompanied by delocalized pi-electron clouds in place of individual alternating double and single bonds. They are also called aromatics or arenes. The best examples are toluene and benzene.
Antiaromatic compounds are not necessarily unstable - they are just less stable than a bunch of ethenes connected by sigma bonds.
Another method to approach these types of questions are:
A molecule is aromatic if it is cyclic, planar, completely conjugated compound with $ 4n + 2\pi $ electrons. It is antiaromatic if all of this is correct except it has 4n electrons, any deviation from these criteria makes it non-aromatic.
Complete step by step answer:
We will start by mentioning the aromaticity rule.
This rule is followed by aromatic and anti- aromatic compounds.
First, the compound to be observed must be cyclic. Also, every atom in the ring must be conjugated. Then the molecule must have $ \left( {4n + 2} \right)\pi $ electrons. At last, the molecule must be flat, but there are rare exceptions.
Option A is cyclooctatriene which is a non-aromatic compound. It changes to tub shaped structure.
Option B is cyclobutadiene which has $ {\text{2 \times n}}{{\text{e}}^{\text{ - }}}{\text{ where n = 2 = 4}}{{\text{e}}^{\text{ - }}} $ ,so it is antiaromatic.
Option C is not planar as the two middle C atoms are $ s{p^3} $ hybridised, thus making it non planar.
Option D is pentagon with negative charge and it is aromatic as it has $ {\text{2n}}{{\text{e}}^{\text{ - }}}{\text{ where n = 3}} $ .
Now we need to select the correct option.
Thus, the correct option is B.
Note:
Aromatic compounds are chemical compounds that consist of conjugated planar ring systems accompanied by delocalized pi-electron clouds in place of individual alternating double and single bonds. They are also called aromatics or arenes. The best examples are toluene and benzene.
Antiaromatic compounds are not necessarily unstable - they are just less stable than a bunch of ethenes connected by sigma bonds.
Another method to approach these types of questions are:
A molecule is aromatic if it is cyclic, planar, completely conjugated compound with $ 4n + 2\pi $ electrons. It is antiaromatic if all of this is correct except it has 4n electrons, any deviation from these criteria makes it non-aromatic.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Which among the following are examples of coming together class 11 social science CBSE




