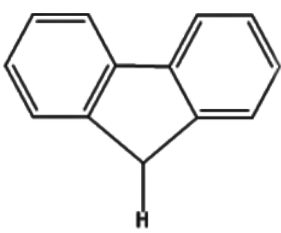
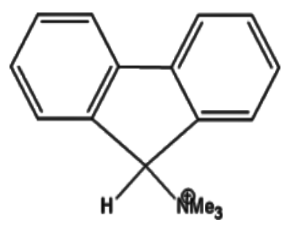
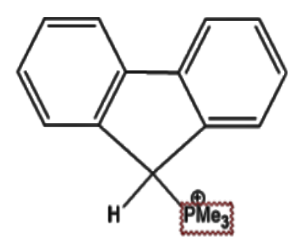
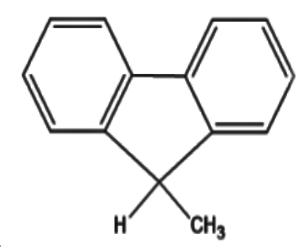
Which of the following has the most acidic hydrogen?
A.
B.
C.
D.




Answer
514.5k+ views
Hint: The most common understanding is that strong acids have relatively stable conjugate bases.The more stable the base stronger will be the acid thus formed.Although the examination of $pKa$ values offers insights into the acidic trends.By using theoretical guidelines too we can estimate the acidity of compounds.
Complete answer: 1)The acidity of hydrogen atoms can be determined with the formation of a stable base.
2)For cyclic compounds the acidity of hydrogen atoms is determined with the stability of resonance hybrids which will eventually form.
3)The presence of strong electron withdrawing groups in the cyclic ring will further stabilise the compound.
4)In all of the options,option (a) lacks any electron withdrawing group attached to the carbon atom which has the hydrogen atom.Hence this option is incorrect.
5)In option (d) the methyl group present is not a strong electron withdrawing group as compared to the groups present in options (b) and option (c).Thus we can safely conclude that option (b) is incorrect.
6)Thus we are left with two options.For this we need to analyse the electron withdrawing capacity of these two groups.
7)The electron withdrawing group $\mathop N\limits^ \oplus M{e_3}$ is a stronger electron withdrawing group as compared to $\mathop P\limits^ \oplus M{e_3}$ hence it will produce a more acidic hydrogen as compared to the latter.This is also due to the fact that $\mathop N\limits^ \oplus M{e_3}$ will aid in the stability of the resonance hybrid which will eventually form.
With these points in consideration we conclude that option (B) is correct.
Note:
There are various factors which we need to consider for the formation of acidic hydrogen. Factors like stability of conjugate base,presence of electron withdrawing groups affect the formation of acidic hydrogen greatly.
Complete answer: 1)The acidity of hydrogen atoms can be determined with the formation of a stable base.
2)For cyclic compounds the acidity of hydrogen atoms is determined with the stability of resonance hybrids which will eventually form.
3)The presence of strong electron withdrawing groups in the cyclic ring will further stabilise the compound.
4)In all of the options,option (a) lacks any electron withdrawing group attached to the carbon atom which has the hydrogen atom.Hence this option is incorrect.
5)In option (d) the methyl group present is not a strong electron withdrawing group as compared to the groups present in options (b) and option (c).Thus we can safely conclude that option (b) is incorrect.
6)Thus we are left with two options.For this we need to analyse the electron withdrawing capacity of these two groups.
7)The electron withdrawing group $\mathop N\limits^ \oplus M{e_3}$ is a stronger electron withdrawing group as compared to $\mathop P\limits^ \oplus M{e_3}$ hence it will produce a more acidic hydrogen as compared to the latter.This is also due to the fact that $\mathop N\limits^ \oplus M{e_3}$ will aid in the stability of the resonance hybrid which will eventually form.
With these points in consideration we conclude that option (B) is correct.
Note:
There are various factors which we need to consider for the formation of acidic hydrogen. Factors like stability of conjugate base,presence of electron withdrawing groups affect the formation of acidic hydrogen greatly.
Recently Updated Pages
Master Class 10 Computer Science: Engaging Questions & Answers for Success

Master Class 10 General Knowledge: Engaging Questions & Answers for Success

Master Class 10 English: Engaging Questions & Answers for Success

Master Class 10 Social Science: Engaging Questions & Answers for Success

Master Class 10 Maths: Engaging Questions & Answers for Success

Master Class 10 Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




