Which of the following cannot be a base?
A.
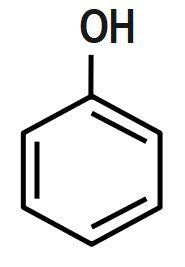
B.
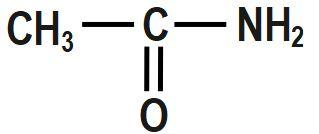
C.
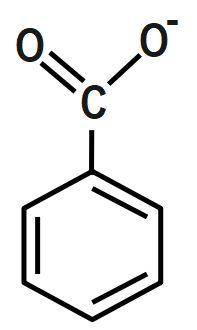
D.
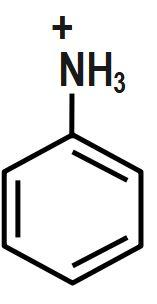

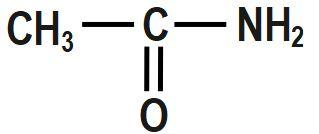


Answer
543k+ views
Hint: We know that we know that base is a substance, which could neutralize acid. When an acid and base react together, the product formed will be salt and water. We can classify base into two types: Strong base and weak base.
Complete answer:
Bases that are water-soluble are called alkali. Both alkali and bases are made up of metal oxides, metal hydroxides, metal carbonates. A base is a substance, which neutralizes an acid. Base has the ability to accept a proton from an acid. Some of the bases such as metal hydroxides and metal oxide form neutral products on reaction with acids. Base is insoluble in water. Salts and water are produced when the base reacts with acids. They have bitter taste and are slippery. There are two types of bases: strong and weak bases.
Bases accept protons and form stable conjugate acids. By observation, we can say that in Option D, there is already a positive charge over the electronegative N-atom. Hence, it will be reluctant to accept any more protons. Thus, it cannot act as a base.
Therefore, the correct answer is option D.
Note:
Remember that the alkalis are bases (or) hydroxide that belongs to the s-block element that is alkali metal and alkali earth metal. Alkali has the ability to accept a proton, and gives out hydroxide ion $\left( O{{H}^{-}} \right)$ . Some of the applications of alkali are, Sodium hydroxide is used in the manufacture of paper, detergents and soaps.
Complete answer:
Bases that are water-soluble are called alkali. Both alkali and bases are made up of metal oxides, metal hydroxides, metal carbonates. A base is a substance, which neutralizes an acid. Base has the ability to accept a proton from an acid. Some of the bases such as metal hydroxides and metal oxide form neutral products on reaction with acids. Base is insoluble in water. Salts and water are produced when the base reacts with acids. They have bitter taste and are slippery. There are two types of bases: strong and weak bases.
Bases accept protons and form stable conjugate acids. By observation, we can say that in Option D, there is already a positive charge over the electronegative N-atom. Hence, it will be reluctant to accept any more protons. Thus, it cannot act as a base.
Therefore, the correct answer is option D.
Note:
Remember that the alkalis are bases (or) hydroxide that belongs to the s-block element that is alkali metal and alkali earth metal. Alkali has the ability to accept a proton, and gives out hydroxide ion $\left( O{{H}^{-}} \right)$ . Some of the applications of alkali are, Sodium hydroxide is used in the manufacture of paper, detergents and soaps.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE




