Which of the following biphenyl is optically active?
(A)
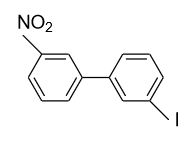
(B)
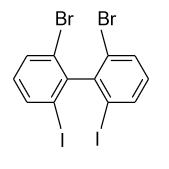
(C)
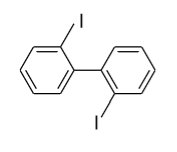
(D)
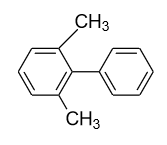




Answer
579.6k+ views
Hint: Optical activity is shown by compounds in which plane of symmetry is not present, if plane of symmetry is present in any compound then they will never show optical activity.
Complete answer:
Optical activity is a kind of property of any compound which rotates the plane polarized light to the right or left side from the direction of upcoming light.
In biphenyls two benzene rings are present and if a plane of symmetry is present within the molecule then they don't show optical activity.
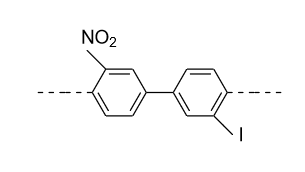
- In option (A), in the 1ST ring nitro (${\text{N}}{{\text{O}}_{\text{2}}}$) group is present in the meta (m) position and in the 2nd ring iodine (${\text{I}}$) is present in the meta’ (m’) position so plane of symmetry is present in this biphenyl in the following manner and it doesn’t show optical activity.
- In option (B), in the ortho positions of both the rings bromine (${\text{Br}}$) atoms are present and in the ortho’ positions iodine (${\text{I}}$) atoms are present. Due to presence of substitution in both the rings at ortho position strain is produced and the compound will not present no more planar in structure. So, there is no plane of symmetry existing and the compound will not show optical activity.
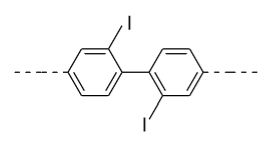
- In option (C), in the 1ST ring iodine (${\text{I}}$) group is present in the ortho (o) position and in the 2nd ring iodine (${\text{I}}$) is present in the ortho’ (o’) position so plane of symmetry is present in this biphenyl in the following manner and it doesn’t show optical activity.
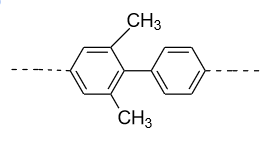
- In option (D), in the 1ST ring methyl group (${\text{C}}{{\text{H}}_{\text{3}}}$) group is present in the ortho (o) and ortho’ (o’) position. So, a plane of symmetry is present in this biphenyl in the following manner and it doesn’t show optical activity.
Hence, option (B) is correct.
Note:
Here some of you may get confused on getting the planarity or non – planarity of the molecule. And for rejecting this type of confusion you should know about the nature of the substituted species at different positions.
Complete answer:
Optical activity is a kind of property of any compound which rotates the plane polarized light to the right or left side from the direction of upcoming light.
In biphenyls two benzene rings are present and if a plane of symmetry is present within the molecule then they don't show optical activity.
- In option (A), in the 1ST ring nitro (${\text{N}}{{\text{O}}_{\text{2}}}$) group is present in the meta (m) position and in the 2nd ring iodine (${\text{I}}$) is present in the meta’ (m’) position so plane of symmetry is present in this biphenyl in the following manner and it doesn’t show optical activity.

- In option (B), in the ortho positions of both the rings bromine (${\text{Br}}$) atoms are present and in the ortho’ positions iodine (${\text{I}}$) atoms are present. Due to presence of substitution in both the rings at ortho position strain is produced and the compound will not present no more planar in structure. So, there is no plane of symmetry existing and the compound will not show optical activity.
- In option (C), in the 1ST ring iodine (${\text{I}}$) group is present in the ortho (o) position and in the 2nd ring iodine (${\text{I}}$) is present in the ortho’ (o’) position so plane of symmetry is present in this biphenyl in the following manner and it doesn’t show optical activity.

- In option (D), in the 1ST ring methyl group (${\text{C}}{{\text{H}}_{\text{3}}}$) group is present in the ortho (o) and ortho’ (o’) position. So, a plane of symmetry is present in this biphenyl in the following manner and it doesn’t show optical activity.

Hence, option (B) is correct.
Note:
Here some of you may get confused on getting the planarity or non – planarity of the molecule. And for rejecting this type of confusion you should know about the nature of the substituted species at different positions.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Trending doubts
Explain zero factorial class 11 maths CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

What is the atomic mass of Sodium a 22 gmol b 23 gmol class 11 chemistry CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

How do I get the molar mass of urea class 11 chemistry CBSE

What organs are located on the left side of your body class 11 biology CBSE




